Disruption of T helper 2-immune responses in Epstein-Barr virus-induced gene 3-deficient mice
- PMID: 12482940
- PMCID: PMC139250
- DOI: 10.1073/pnas.252648899
Disruption of T helper 2-immune responses in Epstein-Barr virus-induced gene 3-deficient mice
Abstract
Epstein-Barr virus-induced gene 3 (EBI3) is a widely expressed IL-12p40-related protein that associates as a heterodimer with either IL-12p35 or an IL-12p35 homologue, p28, to create a new cytokine (IL-27). To define the function of EBI3 in vivo, we generated knockout mice in which the ebi3 gene was targeted by homologous recombination. EBI3-/- mice exhibited normal numbers of both naive and mature CD4+ and CD8+ T cells and B cells, but markedly decreased numbers of invariant natural killer T cells (iNKT) as defined by staining with an alpha-galactosylceramide (alphaGalCer)-loaded CD1d-tetramer. iNKT cells from EBI3-/- mice exhibited decreased IL-4 and, to a lesser extent, IFN-gamma production after alphaGalCer stimulation in vitro. A sustained decrease in IL-4 production was also observed in EBI3-/- mice after alphaGalCer stimulation in vivo in contrast to IFN-gamma production, which was only transiently decreased under such stimulation. Notably, EBI3-/- mice were resistant to the induction of immunopathology associated with oxazolone-induced colitis, a colitis model mediated primarily by T helper (Th) 2-type cytokine production by iNKT cells. In contrast, trinitrobenzene sulfonic acid-induced colitis, a predominantly Th1-mediated colitis model, was unaffected. Thus, EBI3 plays a critical regulatory role in the induction of Th2-type immune responses and the development of Th2-mediated tissue inflammation in vivo, which may be mediated through the control of iNKT cell function.
Figures

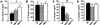
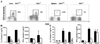

Similar articles
-
Epstein-Barr virus-induced gene 3 suppresses T helper type 1, type 17 and type 2 immune responses after Trypanosoma cruzi infection and inhibits parasite replication by interfering with alternative macrophage activation.Immunology. 2016 Mar;147(3):338-48. doi: 10.1111/imm.12565. Immunology. 2016. PMID: 26694585 Free PMC article.
-
EBI3 deficiency leads to diminished T helper type 1 and increased T helper type 2 mediated airway inflammation.Immunology. 2011 Apr;132(4):559-66. doi: 10.1111/j.1365-2567.2010.03401.x. Epub 2011 Jan 24. Immunology. 2011. PMID: 21255010 Free PMC article.
-
Production of both IL-27 and IFN-gamma after the treatment with a ligand for invariant NK T cells is responsible for the suppression of Th2 response and allergic inflammation in a mouse experimental asthma model.J Immunol. 2009 Jul 1;183(1):254-60. doi: 10.4049/jimmunol.0800520. J Immunol. 2009. PMID: 19542437
-
A Chaperone-Like Role for EBI3 in Collaboration With Calnexin Under Inflammatory Conditions.Front Immunol. 2021 Sep 17;12:757669. doi: 10.3389/fimmu.2021.757669. eCollection 2021. Front Immunol. 2021. PMID: 34603342 Free PMC article. Review.
-
Expanding Diversity in Molecular Structures and Functions of the IL-6/IL-12 Heterodimeric Cytokine Family.Front Immunol. 2016 Nov 4;7:479. doi: 10.3389/fimmu.2016.00479. eCollection 2016. Front Immunol. 2016. PMID: 27867385 Free PMC article. Review.
Cited by
-
Insights from advances in research of chemically induced experimental models of human inflammatory bowel disease.World J Gastroenterol. 2007 Nov 14;13(42):5581-93. doi: 10.3748/wjg.v13.i42.5581. World J Gastroenterol. 2007. PMID: 17948932 Free PMC article. Review.
-
Novel IL-12 family members shed light on the orchestration of Th1 responses.Trends Immunol. 2003 Apr;24(4):207-12. doi: 10.1016/s1471-4906(03)00067-x. Trends Immunol. 2003. PMID: 12697453 Free PMC article. Review.
-
A novel role for interleukin-27 (IL-27) as mediator of intestinal epithelial barrier protection mediated via differential signal transducer and activator of transcription (STAT) protein signaling and induction of antibacterial and anti-inflammatory proteins.J Biol Chem. 2012 Jan 2;287(1):286-298. doi: 10.1074/jbc.M111.294355. Epub 2011 Nov 8. J Biol Chem. 2012. PMID: 22069308 Free PMC article.
-
Epstein-Barr virus-induced gene 3 (EBI3) can mediate IL-6 trans-signaling.J Biol Chem. 2017 Apr 21;292(16):6644-6656. doi: 10.1074/jbc.M116.762021. Epub 2017 Mar 9. J Biol Chem. 2017. PMID: 28280243 Free PMC article.
-
Oral delivery of IL-27 recombinant bacteria attenuates immune colitis in mice.Gastroenterology. 2014 Jan;146(1):210-221.e13. doi: 10.1053/j.gastro.2013.09.060. Epub 2013 Oct 9. Gastroenterology. 2014. PMID: 24120477 Free PMC article.
References
-
- Thierfelder W. E., van Deursen, J. M., Yamamoto, K., Tripp, R. A., Sarawar, S. R., Carson, R. T., Sangster, M. Y., Vignali, D. A., Doherty, P. C., Grosveld, G. C. & Ihle, J. N. (1996) Nature 382, 171-174. - PubMed
-
- Asnagli H. & Murphy, K. M. (2001) Curr. Opin. Immunol. 13, 242-247. - PubMed
-
- Tew J. G., Wu, J., Fakher, M., Szakal, A. K. & Qin, D. (2001) Trends Immunol. 22, 361-367. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

