Bursting in leech heart interneurons: cell-autonomous and network-based mechanisms
- PMID: 12486150
- PMCID: PMC6758431
- DOI: 10.1523/JNEUROSCI.22-24-10580.2002
Bursting in leech heart interneurons: cell-autonomous and network-based mechanisms
Abstract
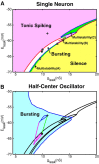
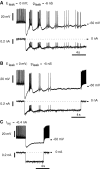
Rhythmic activity within the heartbeat pattern generator of the medicinal leech is based on the alternating bursting of mutually inhibitory pairs of oscillator heart interneurons (half-center oscillators). Bicuculline methiodide has been shown to block mutual inhibition between these interneurons and to cause them to spike tonically while recorded intracellularly (Schmidt and Calabrese, 1992). Using extracellular recording techniques, we show here that oscillator and premotor heart interneurons continue to burst when pharmacologically isolated with bicuculline, although the bursting is not robust in some preparations. We propose that a nonspecific leak current introduced by the intracellular microelectrode suppresses endogenous bursting activity to account for the discrepancy with results using intracellular recording. A two-parameter bifurcation diagram (E(leak) vs g(leak)) of a mathematical model of a single heart interneuron shows a narrow stripe of parameter values where bursting occurs, separating large zones of tonic spiking and silence. A similar analysis performed for a half-center oscillator model outlined a much larger area of bursting. Bursting in the half-center oscillator model is also less sensitive to variation in the maximal conductances of voltage-gated currents than in the single-neuron model. Thus, in addition to ensuring appropriate bursting characteristics such as period, phase, and duty cycles, the half-center configuration enhances oscillation robustness, making them less susceptible to random or imposed changes in membrane parameters. Endogenous bursting, in turn, ensures appropriate bursting if the strength of mutual inhibition is weakened and limits the minimum period of the half-center oscillator to a period near that of the single neuron.
Figures








References
-
- Angstadt JD, Friesen WO. Synchronized oscillatory activity in leech neurons induced by calcium channel blockers. J Neurophysiol. 1991;66:1858–1873. - PubMed
-
- Arbas EA, Calabrese RL. Rate modification in the heartbeat central pattern generator of the medicinal leech. J Comp Physiol [A] 1984;155:783–794.
-
- Arbas EA, Calabrese RL. Leydig neuron activity modulates heartbeat in the medicinal leech. J Comp Physiol [A] 1990;167:665–671. - PubMed
-
- Arshavsky YI, Deliagina TG, Orlovsky GN, Panchin YV, Pavlova GA, Popova LB. Control of locomotion in marine mollusc Clione limacina. VI. Activity of isolated neurons of pedal ganglia. Exp Brain Res. 1986;63:106–112. - PubMed
-
- Arshavsky YI, Orlovsky GN, Panchin YV, Roberts A, Soffe SR. Neuronal control of swimming locomotion: analysis of the pteropod mollusc Clione and embryos of the amphibian Xenopus. Trends Neurosci. 1993;16:227–233. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
