Correlated calcium uptake and release by mitochondria and endoplasmic reticulum of CA3 hippocampal dendrites after afferent synaptic stimulation
- PMID: 12486158
- PMCID: PMC6758462
- DOI: 10.1523/JNEUROSCI.22-24-10653.2002
Correlated calcium uptake and release by mitochondria and endoplasmic reticulum of CA3 hippocampal dendrites after afferent synaptic stimulation
Abstract
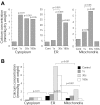
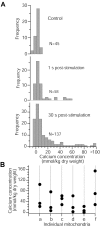
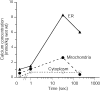
Mitochondria and endoplasmic reticulum (ER) are important modulators of intracellular calcium signaling pathways, but the role of these organelles in shaping synaptic calcium transients in dendrites of pyramidal neurons remains speculative. We have measured directly the concentrations of total Ca (bound plus free) within intracellular compartments of proximal dendrites of CA3 hippocampal neurons at times after synaptic stimulation corresponding to the peak of the cytoplasmic free Ca2+ transient (1 sec), to just after its decay (30 sec), and to well after its return to prestimulus levels (180 sec). Electron probe microanalysis of cryosections from rapidly frozen slice cultures has revealed that afferent mossy fiber stimulation evokes large, rapid elevations in the concentration of total mitochondrial Ca ([Ca](mito)) in depolarized dendrites. A single tetanus (50 Hz/1 sec) elevated [Ca](mito) more than fivefold above characteristically low basal levels within 1 sec of stimulation and >10-fold by 30 sec after stimulation. This strong Ca accumulation was reversible, because [Ca](mito) had recovered by 180 sec after the tetanus. Ca sequestered within mitochondria was localized to small inclusions that were distributed heterogeneously within, and probably among, individual mitochondria. By 30 sec after stimulation an active subpopulation of ER cisterns had accumulated more Ca than had mitochondria despite a approximately 1 sec delay before the onset of accumulation. Active ER cisterns retained their Ca load much longer (>3 min) than mitochondria. The complementary time courses of mitochondrial versus ER Ca2+ uptake and release suggest that these organelles participate in a choreographed interplay, each shaping dendritic Ca2+ signals within characteristic regimes of cytosolic Ca2+ concentration and time.
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous
