Functional and biochemical analysis of a sodium channel beta1 subunit mutation responsible for generalized epilepsy with febrile seizures plus type 1
- PMID: 12486163
- PMCID: PMC6758463
- DOI: 10.1523/JNEUROSCI.22-24-10699.2002
Functional and biochemical analysis of a sodium channel beta1 subunit mutation responsible for generalized epilepsy with febrile seizures plus type 1
Abstract
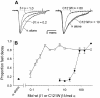
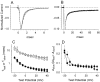
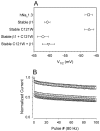
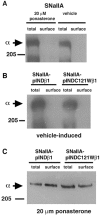
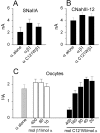
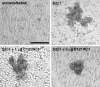
Generalized epilepsy with febrile seizures plus type 1 is an inherited human epileptic syndrome, associated with a cysteine-to-tryptophan (C121W) mutation in the extracellular immunoglobin domain of the auxiliary beta1 subunit of the voltage-gated sodium channel. The mutation disrupts beta1 function, but how this leads to epilepsy is not understood. In this study, we make several observations that may be relevant for understanding why this beta1 mutation results in seizures. First, using electrophysiological recordings from mammalian cell lines, coexpressing sodium channel alpha subunits and either wild-type beta1 or C121Wbeta1, we show that loss of beta1 functional modulation, caused by the C121W mutation, leads to increased sodium channel availability at hyperpolarized membrane potentials and reduced sodium channel rundown during high-frequency channel activity, compared with channels coexpressed with wild-type beta1. In contrast, neither wild-type beta1 nor C121Wbeta1 significantly affected sodium current time course or the voltage dependence of channel activation. We also show, using a Drosophila S2 cell adhesion assay, that the C121W mutation disrupts beta1-beta1 homophilic cell adhesion, suggesting that the mutation may alter the ability of beta1 to mediate protein-protein interactions critical for sodium channel localization. Finally, we demonstrate that neither functional modulation nor cell adhesion mediated by wild-type beta1 is occluded by coexpression of C121Wbeta1, arguing against the idea that the mutant beta1 acts as a dominant-negative subunit. Together, these data suggest that C121Wbeta1 causes subtle effects on channel function and subcellular distribution that bias neurons toward hyperexcitabity and epileptogenesis.
Figures






References
-
- Abou-Khalil B, Ge Q, Desai R, Ryther R, Bazyk A, Baily R, Haines JL, Sutcliff JS, George AL., Jr Partial and generalized epilepsy with febrile seizures plus and a novel SCN1A mutation. Neurology. 2001;57:2265–2272. - PubMed
-
- Alekov AK, Rahman M, Mitrovic N, Lehmann-Horn F, Lerche H. Enhanced inactivation and acceleration of activation of the sodium channel associated with epilepsy in man. Eur J Physiol. 2001;13:2171–2176. - PubMed
-
- Barek S. Site-directed mutagenesis by double polymerase chain reaction: megaprimer method. In: White BA, editor. Methods in molecular biology, Vol 15: PCR protocols, current methods and applications. Humana; Totowa, NJ: 1993. pp. 277–286. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
