Lipooligosaccharide-deficient Neisseria meningitidis shows altered pilus-associated characteristics
- PMID: 12496161
- PMCID: PMC143165
- DOI: 10.1128/IAI.71.1.155-162.2003
Lipooligosaccharide-deficient Neisseria meningitidis shows altered pilus-associated characteristics
Abstract
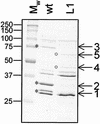
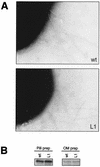
Molecular interaction between host mucosal surfaces and outer membrane components of microbes is crucial in the infection process. The outer membrane of pathogenic Neisseria contains surface molecules such as pili, PilC, and Opa and a monolayer of lipooligosaccharide (LOS), all of which are involved in the interaction with host cells. Pili mediate the initial attachment to human epithelial cells, which is followed by tight contact between bacteria and the eucaryotic cells, leading to bacterial invasion. To further examine the basis for bacterium-host cell contact, we constructed an LOS-deficient Neisseria meningitidis serogroup C mutant. LOS deficiency was without exception accompanied by altered colony opacity and morphology, which most likely represented an "on" switch for Opa540 expression, and by reduced levels of the iron-regulated proteins FetA and FbpA. We show here that LOS is essential for pilus-associated adherence but dispensable for fiber formation and twitching motility. The absence of attachment to epithelial cells could not be attributed to altered levels of piliation or defects in the pilus adhesion phenotype. Further, LOS mutants do not invade host cells and have lost the natural competence for genetic transformation.
Figures

References
-
- Billker, O., A. Popp, S. D. Gray-Owen, and T. F. Meyer. 2000. The structural basis of CEACAM-receptor targeting by neisserial Opa proteins. Trends Microbiol. 8:258-260. - PubMed
-
- Carson, S. D., B. Stone, M. Beucher, J. Fu, and P. F. Sparling. 2000. Phase variation of the gonococcal siderophore receptor FetA. Mol. Microbiol. 36:585-593. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

