Impact of vector priming on the immunogenicity of recombinant Salmonella vaccines
- PMID: 12496178
- PMCID: PMC143414
- DOI: 10.1128/IAI.71.1.287-297.2003
Impact of vector priming on the immunogenicity of recombinant Salmonella vaccines
Abstract
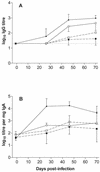
There are conflicting reports concerning the impact of prior vector priming on the immunogenicity of recombinant-Salmonella-based vaccines. A comparison of experimental protocols identified two variables which might account for this inconsistency: the potential of the vector strain to colonize the murine gut-associated lymphoid tissue (GALT) and the nature of the foreign antigen subsequently delivered by the recombinant Salmonella construct. The former was investigated by constructing an aroA mutant of the Salmonella enterica serovar Stanley vector previously used in our laboratory. Although the introduction of an aroA mutation had surprisingly little effect on GALT colonization, it did reduce the strength of antilipopolysaccharide (anti-LPS) antibody responses and the impact of vector priming. Studies were also performed to ascertain the extent to which any observed hyporesponsiveness consequent upon vector priming might be determined by the characteristics of the foreign antigen. S. enterica serovar Stanley was used to deliver either of two Escherichia coli antigens, K88 pilus protein or the LT-B toxin subunit, to vector-primed mice. Both serum immunoglobulin G (IgG) and intestinal IgA responses to K88 were completely abolished, and those to LT-B were significantly reduced, as a consequence of vector priming. When similar experiments were performed with an aroA S. enterica serovar Dublin vector, responses to K88 were significantly reduced but those to LT-B were unaffected by vector priming. Paradoxically, a priming infection with this vector induced stronger anti-LPS antibody responses but was less likely to elicit a state of hyporesponsiveness to subsequently presented foreign antigen. The impact of vector priming thus depends on both the Salmonella strain used and the nature of the foreign antigen, but our present data strengthen concerns that preexisting antivector immunity represents a serious threat to the Salmonella-based vaccine strategy.
Figures



Similar articles
-
Vector-primed mice display hypo-responsiveness to foreign antigen presented by recombinant Salmonella regardless of the route of delivery.Microb Pathog. 2005 Jul-Aug;39(1-2):1-7. doi: 10.1016/j.micpath.2005.05.004. Microb Pathog. 2005. PMID: 16002258
-
Oral delivery of foreign antigens by attenuated Salmonella: consequences of prior exposure to the vector strain.Vaccine. 1997 Feb;15(2):155-62. doi: 10.1016/s0264-410x(96)00158-2. Vaccine. 1997. PMID: 9066032
-
Mucosal and systemic immune responses to chimeric fimbriae expressed by Salmonella enterica serovar typhimurium vaccine strains.Infect Immun. 2000 Jun;68(6):3129-39. doi: 10.1128/IAI.68.6.3129-3139.2000. Infect Immun. 2000. PMID: 10816454 Free PMC article.
-
Live oral Salmonella vaccines: potential use of attenuated strains as carriers of heterologous antigens to the immune system.Parasite Immunol. 1987 Mar;9(2):151-60. doi: 10.1111/j.1365-3024.1987.tb00496.x. Parasite Immunol. 1987. PMID: 3106921 Review.
-
Recombinant Salmonella vectors in vaccine development.Dev Biol Stand. 1994;82:23-33. Dev Biol Stand. 1994. PMID: 7958478 Review.
Cited by
-
Enteric pathogens as vaccine vectors for foreign antigen delivery.Infect Immun. 2004 Oct;72(10):5535-47. doi: 10.1128/IAI.72.10.5535-5547.2004. Infect Immun. 2004. PMID: 15385450 Free PMC article. Review. No abstract available.
-
A live attenuated Listeria monocytogenes vaccine vector expressing SIV Gag is safe and immunogenic in macaques and can be administered repeatedly.Vaccine. 2011 Jan 10;29(3):476-86. doi: 10.1016/j.vaccine.2010.10.072. Epub 2010 Nov 9. Vaccine. 2011. PMID: 21070847 Free PMC article.
-
Strategies for Enhancement of Live-Attenuated Salmonella-Based Carrier Vaccine Immunogenicity.Vaccines (Basel). 2021 Feb 17;9(2):162. doi: 10.3390/vaccines9020162. Vaccines (Basel). 2021. PMID: 33671124 Free PMC article. Review.
-
Vector priming reduces the immunogenicity of Salmonella-based vaccines in Nramp1+/+ mice.Infect Immun. 2003 Apr;71(4):2258-61. doi: 10.1128/IAI.71.4.2258-2261.2003. Infect Immun. 2003. PMID: 12654852 Free PMC article.
-
Live bacterial vaccine vectors: an overview.Braz J Microbiol. 2015 Mar 4;45(4):1117-29. doi: 10.1590/s1517-83822014000400001. eCollection 2014. Braz J Microbiol. 2015. PMID: 25763014 Free PMC article. Review.
References
-
- Attridge, S. R., R. Davies, and J. T. LaBrooy. 1997. Oral delivery of foreign antigens by attenuated Salmonella: consequences of prior exposure to the vector strain. Vaccine 15:155-162. - PubMed
-
- Dougan, G. 1994. The molecular basis for the virulence of bacterial pathogens: implications for oral vaccine development. Microbiology 140:215-224. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

