Roles of the Maltese cross form in the development of parasitemia and protection against Babesia microti infection in mice
- PMID: 12496191
- PMCID: PMC143159
- DOI: 10.1128/IAI.71.1.411-417.2003
Roles of the Maltese cross form in the development of parasitemia and protection against Babesia microti infection in mice
Abstract
Babesia microti, a hemoprotozoan parasite of rodents, is also important as a zoonotic agent of human babesiosis. The Maltese cross form, which consists of four masses in an erythrocyte, is characteristic of the developmental stage of B. microti. Monoclonal antibody (MAb) 2-1E, which specifically recognizes the Maltese cross form of B. microti, has been described previously. In the present study, we examined the roles of the Maltese cross form during the infectious course of B. microti in mice. The number of the Maltese cross form increased in the peripheral blood of infected mice prior to the peak of parasitemia. With confocal laser scanning microscopy, MAb 2-1E was found to be reactive with the ring form, with the parasites undergoing transformation to the Maltese cross form and subsequent division, and also with extracellular merozoites. Furthermore, the Maltese cross form-related antigen (MRA) gene was isolated from a B. microti cDNA library by immunoscreening with MAb 2-1E, and the nucleotide sequence was determined. Genomic analyses indicated that the MRA gene exists as a single-copy gene in B. microti. Immunization of mice with recombinant MRA induced significant protective immunity against B. microti infection. These findings indicate that the Maltese cross form plays important roles in both the development of parasitemia and the protective response against the infection.
Figures
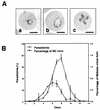



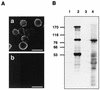


Comment in
-
Roles of the Maltese cross form of Babesia microti in the development of parasitemia in B. microti infection.Infect Immun. 2004 Aug;72(8):4929; author reply 4929-30. doi: 10.1128/IAI.72.8.4929-4930.2004. Infect Immun. 2004. PMID: 15271963 Free PMC article. No abstract available.
References
-
- Avarzed, A., I. Igarashi, D. T. De Waal, S. Kawai, Y. Oomori, N. Inoue, Y. Maki, Y. Omata, A. Saito, H. Nagasawa, Y. Toyoda, and N. Suzuki. 1998. Monoclonal antibody against Babesia equi: characterization and potential application of antigen for serodiagnosis. J. Clin. Microbiol. 36:1835-1839. - PMC - PubMed
-
- Benach, J. L., and G. S. Habicht. 1981. Clinical characteristics of human babesiosis. J. Infect. Dis. 144:481. - PubMed
-
- Fawcett, D. W., P. A. Conrad, J. G. Grootenhuis, and S. P. Morzaria. 1987. Ultrastructure of the intraerythrocytic stage of Theileria species from cattle and waterbuck. Tissue Cell 19:643-655. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
LinkOut - more resources
Full Text Sources

