Combination of drug level measurement and parasite genotyping data for improved assessment of amodiaquine and sulfadoxine-pyrimethamine efficacies in treating Plasmodium falciparum malaria in Gabonese children
- PMID: 12499196
- PMCID: PMC148969
- DOI: 10.1128/AAC.47.1.231-237.2003
Combination of drug level measurement and parasite genotyping data for improved assessment of amodiaquine and sulfadoxine-pyrimethamine efficacies in treating Plasmodium falciparum malaria in Gabonese children
Abstract
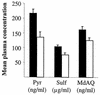
Many African countries currently use a sulfadoxine-pyrimethamine combination (SP) or amodiaquine (AQ) to treat uncomplicated Plasmodium falciparum malaria. Both drugs represent the last inexpensive alternatives to chloroquine. However, resistant P. falciparum populations are largely reported in Africa, and it is compulsory to know the present situation of resistance. The in vivo World Health Organization standard 28-day test was used to assess the efficacy of AQ and SP to treat uncomplicated falciparum malaria in Gabonese children under 10 years of age. To document treatment failures, molecular genotyping to distinguish therapeutic failures from reinfections and drug dosages were undertaken. A total of 118 and 114 children were given AQ or SP, respectively, and were monitored. SP was more effective than AQ, with 14.0 and 34.7% of therapeutic failures, respectively. Three days after initiation of treatment, the mean level of monodesethylamodiaquine (MdAQ) in plasma was 149 ng/ml in children treated with amodiaquine. In those treated with SP, mean levels of sulfadoxine and pyrimethamine in plasma were 100 microg/ml and 212 ng/ml, respectively. Levels of the three drugs were higher in patients successfully treated with AQ (MdAQ plasma levels) or SP (sulfadoxine and pyrimethamine plasma levels). Blood concentration higher than breakpoints of 135 ng/ml for MdAQ, 100 micro g/ml for sulfadoxine, and 175 ng/ml for pyrimethamine were associated with treatment success (odds ratio: 4.5, 9.8, and 11.8, respectively; all P values were <0.009). Genotyping of merozoite surface proteins 1 and 2 demonstrated a mean of 4.0 genotypes per person before treatment. At reappearance of parasitemia, both recrudescent parasites (represented by common bands in both samples) and newly inoculated parasites (represented by bands that were absent before treatment) were present in the blood of most (51.1%) children. Only 3 (6.4%) therapeutic failures were the result not of treatment inefficacy but of new infection. In areas where levels of drug resistance and complexity of infections are high, drug dosage and parasite genotyping may be of limited interest in improving the precision of drug efficacy measurement. Their use should be weighted according to logistical constraints.
Figures
Similar articles
-
Efficacy of chloroquine, amodiaquine and sulphadoxine-pyrimethamine for the treatment of uncomplicated falciparum malaria: revisiting molecular markers in an area of emerging AQ and SP resistance in Mali.Malar J. 2009 Feb 26;8:34. doi: 10.1186/1475-2875-8-34. Malar J. 2009. PMID: 19245687 Free PMC article. Clinical Trial.
-
Artemisinin-based combinations versus amodiaquine plus sulphadoxine-pyrimethamine for the treatment of uncomplicated malaria in Faladje, Mali.Malar J. 2009 Jan 7;8:5. doi: 10.1186/1475-2875-8-5. Malar J. 2009. PMID: 19128455 Free PMC article. Clinical Trial.
-
Low efficacy of amodiaquine or chloroquine plus sulfadoxine-pyrimethamine against Plasmodium falciparum and P. vivax malaria in Papua New Guinea.Am J Trop Med Hyg. 2007 Nov;77(5):947-54. Am J Trop Med Hyg. 2007. PMID: 17984359 Clinical Trial.
-
Chloroquine or amodiaquine combined with sulfadoxine-pyrimethamine for uncomplicated malaria: a systematic review.Trop Med Int Health. 2006 Jun;11(6):789-99. doi: 10.1111/j.1365-3156.2006.01571.x. Trop Med Int Health. 2006. PMID: 16771999
-
Chloroquine or amodiaquine combined with sulfadoxine-pyrimethamine as a treatment for uncomplicated malaria--a systematic review.Ann Trop Med Parasitol. 1998 Apr;92(3):265-70. doi: 10.1080/00034989859825. Ann Trop Med Parasitol. 1998. PMID: 9713541
Cited by
-
Population pharmacokinetics and pharmacodynamic considerations of amodiaquine and desethylamodiaquine in Kenyan adults with uncomplicated malaria receiving artesunate-amodiaquine combination therapy.Antimicrob Agents Chemother. 2010 Jun;54(6):2611-7. doi: 10.1128/AAC.01496-09. Epub 2010 Apr 5. Antimicrob Agents Chemother. 2010. PMID: 20368402 Free PMC article. Clinical Trial.
-
Short course of quinine plus a single dose of sulfadoxine/pyrimethamine for Plasmodium falciparum malaria.Wien Klin Wochenschr. 2006 Oct;118(19-20):610-4. doi: 10.1007/s00508-006-0657-3. Wien Klin Wochenschr. 2006. PMID: 17136336 Clinical Trial.
-
Does the drug sensitivity of malaria parasites depend on their virulence?Malar J. 2008 Dec 16;7:257. doi: 10.1186/1475-2875-7-257. Malar J. 2008. PMID: 19087299 Free PMC article.
-
Sulphadoxine/pyrimethamine versus amodiaquine for treating uncomplicated childhood malaria in Gabon: a randomized trial to guide national policy.Malar J. 2008 Feb 12;7:31. doi: 10.1186/1475-2875-7-31. Malar J. 2008. PMID: 18267042 Free PMC article. Clinical Trial.
-
Carriage of Mutant Dihydrofolate Reductase and Dihydropteroate Synthase Genes among Plasmodium falciparum Isolates Recovered from Pregnant Women with Asymptomatic Infection in Lagos, Nigeria.Med Princ Pract. 2015;24(5):436-43. doi: 10.1159/000430987. Epub 2015 Jun 2. Med Princ Pract. 2015. PMID: 26202938 Free PMC article.
References
-
- Adjuik, M., P. Agnamey, A. Babiker, S. Borrmann, P. Brasseur, M. Cisse, F. Cobelens, S. Diallo, J. F. Faucher, P. Garner, S. Gikunda, P. G. Kremsner, S. Krishna, B. Lell, M. Loolpapit, P. B. Matsiegui, M. A. Missinou, J. Mwanza, F. Ntoumi, P. Olliaro, P. Osimbo, P. Rezbach, E. Some, and W. R. Taylor. 2002. Amodiaquine-artesunate versus amodiaquine for uncomplicated Plasmodium falciparum malaria in African children: a randomized, multicentre trial. Lancet 359:1365-1372. - PubMed
-
- Al-Yaman, F., B. Genton, J. C. Reeder, R. F. Anders, and M. P. Alpers. 1997. Evidence that recurrent Plasmodium falciparum infection is caused by recrudescence of resistant parasites. Am. J. Trop. Med. Hyg. 56:436-439. - PubMed
-
- Basco, L., and P. Ringwald. 2000. Chimiorésistance du paludisme: problèmes de la définition et de l'approche technique. Santé 10:47-50. - PubMed
-
- Basco, L. K., A. Same-Ekobo, V. F. Ngane, M. Ndounga, T. Metoh, P. Ringwald, and G. Soula. 2002. Therapeutic efficacy of sulfadoxine-pyrimethamine, amodiaquine and the sulfadoxine-pyrimethamine-amodiaquine combination against uncomplicated Plasmodium falciparum malaria in young children in Cameroon. Bull. W. H. O. 80:538-545. - PMC - PubMed
-
- Bojang, K. A., G. Schneider, S. Forck, S. K. Obaro, S. Jaffar, M. Pinder, J. Rowley, and B. M. Greenwood. 1998. A trial of Fansidar plus chloroquine or Fansidar alone for the treatment of uncomplicated malaria in Gambian children. Trans. R. Soc. Trop. Med. Hyg. 92:73-76. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

