Metabolism of Cisplatin to a nephrotoxin in proximal tubule cells
- PMID: 12506132
- PMCID: PMC6361148
- DOI: 10.1097/01.asn.0000042803.28024.92
Metabolism of Cisplatin to a nephrotoxin in proximal tubule cells
Abstract
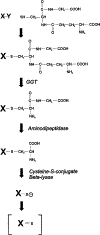
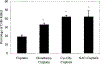
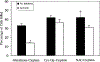
Cisplatin, a commonly used chemotherapeutic agent, is nephrotoxic. The mechanism by which cisplatin selectively kills the proximal tubule cells was heretofore unknown. Recent studies in mice and rats have shown that the nephrotoxicity of cisplatin can be blocked by acivicin or (aminooxy)acetic acid, the same enzyme inhibitors that block the metabolic activation of a series of nephrotoxic halogenated alkenes. In this study, it was hypothesized that cisplatin is activated in the kidney to a toxic metabolite through the same pathway that has been shown to activate the halogenated alkenes. This activation begins with the formation of a glutathione-conjugate that is metabolized to a cysteinyl-glycine-conjugate, to a cysteine-conjugate, and finally to a reactive thiol. In this study, a protocol was developed in which confluent monolayers of LLC-PK(1) cells were exposed to clinically relevant concentrations of cisplatin or cisplatin-conjugate for 3 h. Cell viability was assayed at 72 h. The role of gamma-glutamyl transpeptidase (GGT) and cysteine-S-conjugate beta-lyase in the metabolism of each of the cisplatin-conjugates was investigated. Pre-incubation of cisplatin with glutathione, cysteinyl-glycine, or N-acetyl-cysteine to allow for the spontaneous formation of cisplatin-conjugates increased the toxicity of cisplatin toward LLC-PK(1) cells. Inhibition of GGT activity showed that GGT was necessary only for the toxicity of the cisplatin-glutathione-conjugate. Inhibition of cysteine-S-conjugate beta-lyase reduced the toxicity of each of the cisplatin-conjugates. These data demonstrate that metabolism of cisplatin in proximal tubule cells is required for its nephrotoxicity. The elucidation of this pathway provides new targets for the inhibition of cisplatin nephrotoxicity.
Figures
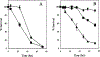
Similar articles
-
Role of cysteine S-conjugate beta-lyase in the metabolism of cisplatin.J Pharmacol Exp Ther. 2003 Sep;306(3):988-94. doi: 10.1124/jpet.103.052225. Epub 2003 May 15. J Pharmacol Exp Ther. 2003. PMID: 12750429
-
High pressure liquid chromatography and mass spectrometry characterization of the nephrotoxic biotransformation products of Cisplatin.Drug Metab Dispos. 2003 Jun;31(6):705-13. doi: 10.1124/dmd.31.6.705. Drug Metab Dispos. 2003. PMID: 12756201 Free PMC article.
-
Inhibition of gamma-glutamyl transpeptidase or cysteine S-conjugate beta-lyase activity blocks the nephrotoxicity of cisplatin in mice.J Pharmacol Exp Ther. 2002 Jan;300(1):142-8. doi: 10.1124/jpet.300.1.142. J Pharmacol Exp Ther. 2002. PMID: 11752109 Free PMC article.
-
Biosynthesis and biotransformation of glutathione S-conjugates to toxic metabolites.Crit Rev Toxicol. 1988;18(4):311-41. doi: 10.3109/10408448809037470. Crit Rev Toxicol. 1988. PMID: 3288445 Review.
-
Cysteine S-conjugate β-lyases: important roles in the metabolism of naturally occurring sulfur and selenium-containing compounds, xenobiotics and anticancer agents.Amino Acids. 2011 Jun;41(1):7-27. doi: 10.1007/s00726-010-0552-0. Epub 2010 Mar 22. Amino Acids. 2011. PMID: 20306345 Free PMC article. Review.
Cited by
-
Cisplatin-induced Kidney Dysfunction and Perspectives on Improving Treatment Strategies.Electrolyte Blood Press. 2014 Dec;12(2):55-65. doi: 10.5049/EBP.2014.12.2.55. Epub 2014 Dec 31. Electrolyte Blood Press. 2014. PMID: 25606044 Free PMC article. Review.
-
A robust, accurate, sensitive LC-MS/MS method to measure indoxyl sulfate, validated for plasma and kidney cells.Biomed Chromatogr. 2022 May;36(5):e5307. doi: 10.1002/bmc.5307. Epub 2022 Feb 8. Biomed Chromatogr. 2022. PMID: 34978088 Free PMC article.
-
Glutathione S-conjugates as prodrugs to target drug-resistant tumors.Front Pharmacol. 2014 Aug 11;5:181. doi: 10.3389/fphar.2014.00181. eCollection 2014. Front Pharmacol. 2014. PMID: 25157234 Free PMC article. Review.
-
High-throughput imaging-based nephrotoxicity prediction for xenobiotics with diverse chemical structures.Arch Toxicol. 2016 Nov;90(11):2793-2808. doi: 10.1007/s00204-015-1638-y. Epub 2015 Nov 27. Arch Toxicol. 2016. PMID: 26612367 Free PMC article.
-
Cisplatin nephrotoxicity: a review of the literature.J Nephrol. 2018 Feb;31(1):15-25. doi: 10.1007/s40620-017-0392-z. Epub 2017 Apr 5. J Nephrol. 2018. PMID: 28382507 Review.
References
-
- Giaccone G: Clinical perspectives on platinum resistance. Drugs 59[Suppl 4]: 9–17, 2000 - PubMed
-
- Pinzani V, Bressolle F, Haug IJ, Galtier M, Blayac JP: Cisplatin induced renal toxicity and toxicity-modulating strategies: A re- view. Cancer Chemother Pharmacol 35: 1–9, 1994 - PubMed
-
- Fink D, Howell SB: How does cisplatin kill cells? In: Platinum-based drugs in cancer therapy edited by Kelland LR, Favilli F, Totowa, New Jersey, Humana Press, 2000, pp 149–167
-
- Lieberthal W, Triaca V, Levine J: Mechanisms of death induced by cisplatin in proximal tubular epithelial cells: apoptosis vs. necrosis. Am J Physiol 270: F700–F708, 1996 - PubMed
-
- Park MS, De Leon M, Devarajan P: Cisplatin induces apoptosis in LLC-PK1 cells via activation of mitochondrial pathways. J Am Soc Nephrol 13: 858–865, 2002 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

