A crucial role of glycoprotein VI for platelet recruitment to the injured arterial wall in vivo
- PMID: 12515812
- PMCID: PMC2193801
- DOI: 10.1084/jem.20020945
A crucial role of glycoprotein VI for platelet recruitment to the injured arterial wall in vivo
Abstract
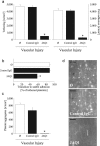
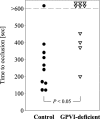
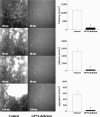
Platelet adhesion and aggregation at sites of vascular injury is crucial for hemostasis but may lead to arterial occlusion in the setting of atherosclerosis and precipitate diseases such as myocardial infarction. A current hypothesis suggests that platelet glycoprotein (GP) Ib interaction with von Willebrand factor recruits flowing platelets to the injured vessel wall, where subendothelial fibrillar collagens support their firm adhesion and activation. However, so far this hypothesis has not been tested in vivo. Here, we demonstrate by intravital fluorescence microscopy of the mouse carotid artery that inhibition or absence of the major platelet collagen receptor, GPVI, abolishes platelet-vessel wall interactions after endothelial denudation. Unexpectedly, inhibition of GPVI by the monoclonal antibody JAQ1 reduced platelet tethering to the subendothelium by approximately 89%. In addition, stable arrest and aggregation of platelets was virtually abolished under these conditions. Using different models of arterial injury, the strict requirement for GPVI in these processes was confirmed in GPVI-deficient mice, where platelets also failed to adhere and aggregate on the damaged vessel wall. These findings reveal an unexpected role of GPVI in the initiation of platelet attachment at sites of vascular injury and unequivocally identify platelet-collagen interactions (via GPVI) as the major determinant of arterial thrombus formation.
Figures







References
-
- Ruggeri, Z.M. 1997. Mechanisms initiating platelet thrombus formation. Thromb. Haemost. 78:611–616. - PubMed
-
- Clemetson, K.J., and J.M. Clemetson. 2001. Platelet collagen receptors. Thromb. Haemost. 86:189–197. - PubMed
-
- Baumgartner, H.R. 1977. Platelet interaction with collagen fibrils in flowing blood. I. Reaction of human platelets with alpha chymotrypsin-digested subendothelium. Thromb. Haemost. 37:1–16. - PubMed
-
- Savage, B., F. Almus-Jacobs, and Z.M. Ruggeri. 1998. Specific synergy of multiple substrate-receptor interactions in platelet thrombus formation under flow. Cell. 94:657–666. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

