Liver sinusoidal endothelial cells represents an important blood clearance system in pigs
- PMID: 12537582
- PMCID: PMC149430
- DOI: 10.1186/1476-5926-2-1
Liver sinusoidal endothelial cells represents an important blood clearance system in pigs
Abstract
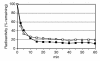
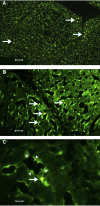
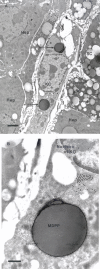
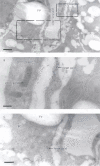
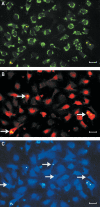
BACKGROUND: Numerous studies in rats and a few other mammalian species, including man, have shown that the sinusoidal cells constitute an important part of liver function. In the pig, however, which is frequently used in studies on liver transplantation and liver failure models, our knowledge about the function of hepatic sinusoidal cells is scarce. We have explored the scavenger function of pig liver sinusoidal endothelial cells (LSEC), a cell type that in other mammals performs vital elimination of an array of waste macromolecules from the circulation. RESULTS: 125I-macromolecules known to be cleared in the rat via the scavenger and mannose receptors were rapidly removed from the pig circulation, 50% of the injected dose being removed within the first 2-5 min following injection. Fluorescently labeled microbeads (2 &mgr;m in diameter) used to probe phagocytosis accumulated in Kupffer cells only, whereas fluorescently labeled soluble macromolecular ligands for the mannose and scavenger receptors were sequestered only by LSEC. Desmin-positive stellate cells accumulated no probes. Isolation of liver cells using collagenase perfusion through the portal vein, followed by various centrifugation protocols to separate the different liver cell populations yielded 280 x 107 (range 50-890 x 107) sinusoidal cells per liver (weight of liver 237.1 g (sd 43.6)). Use of specific anti-Kupffer cell- and anti-desmin antibodies, combined with endocytosis of fluorescently labeled macromolecular soluble ligands indicated that the LSEC fraction contained 62 x 107 (sd 12 x 107) purified LSEC. Cultured LSEC avidly endocytosed ligands for the mannose and scavenger receptors. CONCLUSIONS: We show here for the first time that pig LSEC, similar to what has been found earlier in rat LSEC, represent an effective scavenger system for removal of macromolecular waste products from the circulation.
Figures



References
-
- Watanabe FD, Mullon CJ, Hewitt WR, Arkadopoulos N, Kahaku E, Eguchi S, Khalili T, Arnaout W, Shackleton CR, Rozga J, Solomon B, Demetriou AA. Clinical experience with a bioartificial liver in the treatment of severe liver failure. A phase I clinical trial. Ann Surg. 1997;225:484–491. doi: 10.1097/00000658-199705000-00005. - DOI - PMC - PubMed
-
- Aschoff L. Ergeb Inn Med Kinderhk. 1924. pp. 1–118.
-
- Smedsrød B, Tollersrud OK. Sinusoidal liver endothelial cells recruit lysosomal enzymes from the circulation by mannose-receptor mediated endocytosis. In: Wake K, Wisse E, Knook DL, editor. Cells of the hepatic sinusoid. Leiden, Kupffer Cell Foundation; 1995. pp. 180–183.
LinkOut - more resources
Full Text Sources
Other Literature Sources
