Growth phase-coupled alterations in cell structure and function of Escherichia coli
- PMID: 12562804
- PMCID: PMC142870
- DOI: 10.1128/JB.185.4.1338-1345.2003
Growth phase-coupled alterations in cell structure and function of Escherichia coli
Abstract
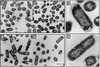
Escherichia coli cultures can be fractionated into more than 20 cell populations, each having a different bouyant density and apparently representing a specific stage of cell differentiation from exponential growth to stationary phase (H. Makinoshima, A. Nishimura, and A. Ishihama, Mol. Microbiol. 43:269-279, 2002). The density increase was found to be impaired at an early step for a mutant E. coli with the disrupted rpoS gene, which encodes the RNA polymerase RpoS (sigma-S) for stationary-phase gene transcription. This finding suggests that RpoS is need for the entire process of cell density increase. In the absence of RpoF sigma factor, the flagella are not formed as observed by electron microscopy, but the growth phase-coupled density increase takes place as in wild-type E. coli, confirming that the alteration in cell density is not directly correlated with the presence or absence of flagella. In the stationary-phase cells, accumulation of electron-dense areas was observed by electron microscopic observation of bacterial thin sections. By chemical determination, the increase in glycogen (or polysaccharides) was suggested to be one component, which contributes to the increase in weight-to-volume ratio of stationary-phase E. coli cells.
Figures





References
-
- Beveridge, T. T., T. J. Popkin, and R. M. Cole. 1994. Electron microscopy, p. 42-71. In P. Gerhardt et al. (ed.), Methods for general and molecular bacteriology. American Society for Microbiology, Washington, D.C.
-
- Claret, L., and C. Hughes. 2002. Interaction of the atypical prokaryotic transcription activator FlhD2C2 with early promoters of the flagellar gene hiererchy. J. Mol. Biol. 321:185-199. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

