Glycerol-3-phosphate acquisition in spirochetes: distribution and biological activity of glycerophosphodiester phosphodiesterase (GlpQ) among Borrelia species
- PMID: 12562805
- PMCID: PMC142843
- DOI: 10.1128/JB.185.4.1346-1356.2003
Glycerol-3-phosphate acquisition in spirochetes: distribution and biological activity of glycerophosphodiester phosphodiesterase (GlpQ) among Borrelia species
Abstract
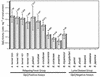
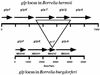
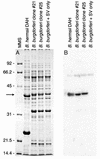
Relapsing-fever spirochetes achieve high cell densities (>10(8)/ml) in their host's blood, while Lyme disease spirochetes do not (<10(5)/ml). This striking contrast in pathogenicity of these two groups of bacteria suggests a fundamental difference in their ability to either exploit or survive in blood. Borrelia hermsii, a tick-borne relapsing-fever spirochete, contains orthologs to glpQ and glpT, genes that encode glycerophosphodiester phosphodiesterase (GlpQ) and glycerol-3-phosphate transporter (GlpT), respectively. In other bacteria, GlpQ hydrolyzes deacylated phospholipids to glycerol-3-phosphate (G3P) while GlpT transports G3P into the cytoplasm. Enzyme assays on 17 isolates of borreliae demonstrated GlpQ activity in relapsing-fever spirochetes but not in Lyme disease spirochetes. Southern blots demonstrated glpQ and glpT in all relapsing-fever spirochetes but not in the Lyme disease group. A Lyme disease spirochete, Borrelia burgdorferi, that was transformed with a shuttle vector containing glpTQ from B. hermsii produced active enzyme, which demonstrated the association of glpQ with the hydrolysis of phospholipids. Sequence analysis of B. hermsii identified glpF, glpK, and glpA, which encode the glycerol facilitator, glycerol kinase, and glycerol-3-phosphate dehydrogenase, respectively, all of which are present in B. burgdorferi. All spirochetes examined had gpsA, which encodes the enzyme that reduces dihydroxyacetone phosphate (DHAP) to G3P. Consequently, three pathways for the acquisition of G3P exist among borreliae: (i) hydrolysis of deacylated phospholipids, (ii) reduction of DHAP, and (iii) uptake and phosphorylation of glycerol. The unique ability of relapsing-fever spirochetes to hydrolyze phospholipids may contribute to their higher cell densities in blood than those of Lyme disease spirochetes.
Figures


References
-
- Barbour, A. G., G. O. Maupin, G. J. Teltow, C. J. Carter, and J. Piesman. 1996. Identification of an uncultivable Borrelia species in the hard tick Amblyomma americanum: possible agent of a Lyme disease-like illness. J. Infect. Dis. 173:403-409. - PubMed
-
- Benach, J. L., E. M. Bosler, J. P. Hanrahan, J. L. Coleman, G. S. Habicht, T. F. Bast, D. J. Cameron, J. L. Ziegler, A. G. Barbour, W. Burgdorfer, R. Edelman, and R. A. Kaslow. 1983. Spirochetes isolated from the blood of two patients with Lyme disease. N. Engl. J. Med. 308:740-742. - PubMed
-
- Bublitz, C., and O. Wieland. 1962. Glycerokinase. Methods Enzymol. 5:354-361.
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

