Biofilm growth and detachment of Actinobacillus actinomycetemcomitans
- PMID: 12562811
- PMCID: PMC142852
- DOI: 10.1128/JB.185.4.1399-1404.2003
Biofilm growth and detachment of Actinobacillus actinomycetemcomitans
Abstract
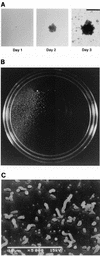
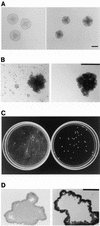
The gram-negative, oral bacterium Actinobacillus actinomycetemcomitans has been implicated as the causative agent of several forms of periodontal disease in humans. When cultured in broth, fresh clinical isolates of A. actinomycetemcomitans form tenacious biofilms on surfaces such as glass, plastic, and saliva-coated hydroxyapatite, a property that probably plays an important role in the ability of this bacterium to colonize the oral cavity and cause disease. We examined the morphology of A. actinomycetemcomitans biofilm colonies grown on glass slides and in polystyrene petri dishes by using light microscopy and scanning and transmission electron microscopy. We found that A. actinomycetemcomitans developed asymmetric, lobed biofilm colonies that displayed complex architectural features, including a layer of densely packed cells on the outside of the colony and nonaggregated cells and large, transparent cavities on the inside of the colony. Mature biofilm colonies released single cells or small clusters of cells into the medium. These released cells adhered to the surface of the culture vessel and formed new colonies, enabling the biofilm to spread. We isolated three transposon insertion mutants which produced biofilm colonies that lacked internal, nonaggregated cells and were unable to release cells into the medium. All three transposon insertions mapped to genes required for the synthesis of the O polysaccharide (O-PS) component of lipopolysaccharide. Plasmids carrying the complementary wild-type genes restored the ability of mutant strains to synthesize O-PS and release cells into the medium. Our findings suggest that A. actinomycetemcomitans biofilm growth and detachment are discrete processes and that biofilm cell detachment evidently involves the formation of nonaggregated cells inside the biofilm colony that are destined for release from the colony.
Figures

Similar articles
-
Detachment of Actinobacillus actinomycetemcomitans biofilm cells by an endogenous beta-hexosaminidase activity.J Bacteriol. 2003 Aug;185(16):4693-8. doi: 10.1128/JB.185.16.4693-4698.2003. J Bacteriol. 2003. PMID: 12896987 Free PMC article.
-
Environmental influences on Actinobacillus actinomycetemcomitans biofilm formation.Arch Oral Biol. 2006 Apr;51(4):299-314. doi: 10.1016/j.archoralbio.2005.09.002. Epub 2005 Oct 13. Arch Oral Biol. 2006. PMID: 16226214
-
Biofilm formation by a fimbriae-deficient mutant of Actinobacillus actinomycetemcomitans.Microbiol Immunol. 2003;47(11):877-81. doi: 10.1111/j.1348-0421.2003.tb03454.x. Microbiol Immunol. 2003. PMID: 14638999
-
Autoinducer 2 is required for biofilm growth of Aggregatibacter (Actinobacillus) actinomycetemcomitans.Infect Immun. 2007 Sep;75(9):4211-8. doi: 10.1128/IAI.00402-07. Epub 2007 Jun 25. Infect Immun. 2007. PMID: 17591788 Free PMC article.
-
Genes for tight adherence of Actinobacillus actinomycetemcomitans: from plaque to plague to pond scum.Trends Microbiol. 2001 Sep;9(9):429-37. doi: 10.1016/s0966-842x(01)02161-8. Trends Microbiol. 2001. PMID: 11553455 Review.
Cited by
-
Should we stay or should we go: mechanisms and ecological consequences for biofilm dispersal.Nat Rev Microbiol. 2011 Nov 28;10(1):39-50. doi: 10.1038/nrmicro2695. Nat Rev Microbiol. 2011. PMID: 22120588 Review.
-
Amylases: Biofilm Inducer or Biofilm Inhibitor?Front Cell Infect Microbiol. 2021 Apr 27;11:660048. doi: 10.3389/fcimb.2021.660048. eCollection 2021. Front Cell Infect Microbiol. 2021. PMID: 33987107 Free PMC article. Review.
-
Role of a nosX homolog in Streptococcus gordonii in aerobic growth and biofilm formation.J Bacteriol. 2004 Dec;186(24):8193-206. doi: 10.1128/JB.186.24.8193-8206.2004. J Bacteriol. 2004. PMID: 15576767 Free PMC article.
-
Surface display of Aggregatibacter actinomycetemcomitans autotransporter Aae and dispersin B hybrid act as antibiofilm agents.Mol Oral Microbiol. 2016 Aug;31(4):329-39. doi: 10.1111/omi.12126. Epub 2015 Sep 22. Mol Oral Microbiol. 2016. PMID: 26280561 Free PMC article.
-
Attenuation of Aggregatibacter actinomycetemcomitans virulence using curcumin-decorated nanophytosomes-mediated photo-sonoantimicrobial chemotherapy.Sci Rep. 2021 Mar 16;11(1):6012. doi: 10.1038/s41598-021-85437-6. Sci Rep. 2021. PMID: 33727630 Free PMC article.
References
-
- Berthold, P., D. Forti, I. R. Kieba, J. Rosenbloom, N. S. Taichman, and E. T. Lally. 1992. Electron immunocytochemical localization of Actinobacillus actinomycetemcomitans leukotoxin. Oral Microbiol. Immunol. 7:24-27. - PubMed
-
- Blix, I. J. S., H. R. Preus, and I. Olsen. 1990. Invasive growth of Actinobacillus actinomycetemcomitans on solid medium (TSBV). Acta Odontol. Scand. 48:313-318. - PubMed
-
- Bowden, M. G., and H. B. Kaplan. 1998. The Myxococcus xanthus lipopolysaccharide O-antigen is required for social motility and multicellular development. Mol. Microbiol. 30:275-284. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

