Beta-oxidation of 5-hydroxydecanoate, a putative blocker of mitochondrial ATP-sensitive potassium channels
- PMID: 12562916
- PMCID: PMC2342646
- DOI: 10.1113/jphysiol.2002.037044
Beta-oxidation of 5-hydroxydecanoate, a putative blocker of mitochondrial ATP-sensitive potassium channels
Abstract
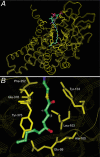
5-Hydroxydecanoate (5-HD) inhibits ischaemic and pharmacological preconditioning of the heart. Since 5-HD is thought to inhibit specifically the putative mitochondrial ATP-sensitive K+ (KATP) channel, this channel has been inferred to be a mediator of preconditioning. However, it has recently been shown that 5-HD is a substrate for acyl-CoA synthetase, the mitochondrial enzyme which 'activates' fatty acids. Here, we tested whether activated 5-HD, 5-hydroxydecanoyl-CoA (5-HD-CoA), is a substrate for medium-chain acyl-CoA dehydrogenase (MCAD), the committed step of the mitochondrial beta-oxidation pathway. Using a molecular model, we predicted that the hydroxyl group on the acyl tail of 5-HD-CoA would not sterically hinder the active site of MCAD. Indeed, we found that 5-HD-CoA was a substrate for purified human liver MCAD with a Km of 12.8 +/- 0.6 microM and a kcat of 14.1 s-1. For comparison, with decanoyl-CoA (Km approximately 3 microM) as substrate, kcat was 6.4 s-1. 5-HD-CoA was also a substrate for purified pig kidney MCAD. We next tested whether the reaction product, 5-hydroxydecenoyl-CoA (5-HD-enoyl-CoA), was a substrate for enoyl-CoA hydratase, the second enzyme of the beta-oxidation pathway. Similar to decenoyl-CoA, purified 5-HD-enoyl-CoA was also a substrate for the hydratase reaction. In conclusion, we have shown that 5-HD is metabolised at least as far as the third enzyme of the beta-oxidation pathway. Our results open the possibility that beta-oxidation of 5-HD or metabolic intermediates of 5-HD may be responsible for the inhibitory effects of 5-HD on preconditioning of the heart.
Figures



Similar articles
-
5-Hydroxydecanoate is metabolised in mitochondria and creates a rate-limiting bottleneck for beta-oxidation of fatty acids.J Physiol. 2005 Jan 15;562(Pt 2):307-18. doi: 10.1113/jphysiol.2004.073932. Epub 2004 Oct 28. J Physiol. 2005. PMID: 15513944 Free PMC article.
-
K(ATP) channel-independent targets of diazoxide and 5-hydroxydecanoate in the heart.J Physiol. 2002 Aug 1;542(Pt 3):735-41. doi: 10.1113/jphysiol.2002.023960. J Physiol. 2002. PMID: 12154175 Free PMC article.
-
5-Hydroxydecanoate and coenzyme A are inhibitors of native sarcolemmal KATP channels in inside-out patches.Biochim Biophys Acta. 2010 Mar;1800(3):385-91. doi: 10.1016/j.bbagen.2009.11.012. Epub 2009 Nov 18. Biochim Biophys Acta. 2010. PMID: 19931596
-
Structure and mechanism of action of the acyl-CoA dehydrogenases.FASEB J. 1995 Jun;9(9):718-25. doi: 10.1096/fasebj.9.9.7601336. FASEB J. 1995. PMID: 7601336 Review.
-
Acyl-CoA dehydrogenases. A mechanistic overview.Eur J Biochem. 2004 Feb;271(3):494-508. doi: 10.1046/j.1432-1033.2003.03946.x. Eur J Biochem. 2004. PMID: 14728676 Review.
Cited by
-
Diazoxide-induced respiratory inhibition - a putative mitochondrial K(ATP) channel independent mechanism of pharmacological preconditioning.Mol Cell Biochem. 2007 Jan;294(1-2):11-8. doi: 10.1007/s11010-005-9066-6. Epub 2006 Nov 29. Mol Cell Biochem. 2007. PMID: 17136444
-
MitoK(ATP)-dependent changes in mitochondrial volume and in complex II activity during ischemic and pharmacological preconditioning of Langendorff-perfused rat heart.J Bioenerg Biomembr. 2006 Apr;38(2):101-12. doi: 10.1007/s10863-006-9016-3. Epub 2006 Sep 21. J Bioenerg Biomembr. 2006. PMID: 17031549
-
KATP Channels in the Cardiovascular System.Physiol Rev. 2016 Jan;96(1):177-252. doi: 10.1152/physrev.00003.2015. Physiol Rev. 2016. PMID: 26660852 Free PMC article. Review.
-
Variable effects of the mitoK(ATP) channel modulators diazoxide and 5-HD in ATP-depleted renal epithelial cells.Mol Cell Biochem. 2010 Feb;335(1-2):211-22. doi: 10.1007/s11010-009-0271-6. Epub 2009 Sep 26. Mol Cell Biochem. 2010. PMID: 19784759
-
Mitochondrial-mediated suppression of ROS production upon exposure of neurons to lethal stress: mitochondrial targeted preconditioning.Adv Drug Deliv Rev. 2008 Oct-Nov;60(13-14):1471-7. doi: 10.1016/j.addr.2008.03.020. Epub 2008 Jul 4. Adv Drug Deliv Rev. 2008. PMID: 18652858 Free PMC article. Review.
References
-
- Chu CH, Kushner L, Cuebas D, Schulz H. The activity of 3-hydroxyacyl-CoA epimerase is insufficient to account for the rate of linoleate oxidation in rat heart mitochondria. Evidence for a modified pathway of linoleate degradation. Biochem Biophys Res Commun. 1984;118:162–167. - PubMed
-
- Cope DK, Impastato WK, Cohen MV, Downey JM. Volatile anesthetics protect the ischemic rabbit myocardium from infarction. Anesthesiology. 1997;86:699–709. - PubMed
-
- Garlid KD, Paucek P, Yarov-Yarovoy V, Murray HN, Darbenzio RB, D'Alonzo AJ, Lodge NJ, Smith MA, Grover GJ. Cardioprotective effect of diazoxide and its interaction with mitochondrial ATP-sensitive K+ channels. Possible mechanism of cardioprotection. Circ Res. 1997;81:1072–1082. - PubMed
-
- Gross GJ. The role of mitochondrial KATP channels in cardioprotection. Basic Res Cardiol. 2000;95:280–284. - PubMed
-
- Gross GJ, Fryer RM. Sarcolemmal versus mitochondrial ATP-sensitive K+ channels and myocardial preconditioning. Circ Res. 1999;84:973–979. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

