A novel chloride conductance activated by extracellular ATP in mouse parotid acinar cells
- PMID: 12562938
- PMCID: PMC2342604
- DOI: 10.1113/jphysiol.2002.028373
A novel chloride conductance activated by extracellular ATP in mouse parotid acinar cells
Abstract
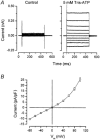
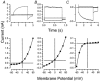
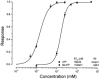
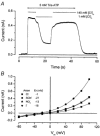
Salivary gland fluid secretion is driven by transepithelial Cl- movement involving an apical Cl- channel whose molecular identity remains unknown. Extracellular ATP (ATP(o)) has been shown to activate a Cl- conductance (I(ATPCl)) in secretory epithelia; to gain further insight into I(ATPCl) in mouse parotid acinar cells, we investigated the effects of ATP(o) using the whole-cell patch-clamp technique. ATP(o) and 2'- and 3'-O-(4-benzoylbenzoyl)adenosine 5'-triphosphate triethylammonium salt (Bz-ATP) produced concentration-dependent, time-independent Cl- currents with an EC50 of 160 and 15 microM, respectively. I(ATPCl) displayed a selectivity sequence of SCN- > I- = NO3- > Cl- > glutamate, similar to the Cl- channels activated by Ca2+, cAMP and cell swelling in acinar cells. In contrast, I(ATPCl) was insensitive to pharmacological agents that are known to inhibit these latter Cl- channels, was independent of Ca2+ and was not regulated by cell volume. Moreover, the I(ATPCl) magnitude from wild-type animals was comparable to that from mice with null mutations in the Cftr, Clcn3 and Clcn2 Cl- channel genes. Taken together, our results demonstrate that I(ATPCl) is distinct from the channels described previously in acinar cells. The activation of I(ATPCl) by Bz-ATP suggests that P2 nucleotide receptors are involved. However, inhibition of G-protein activation with GDP-beta-S failed to block I(ATPCl), and Cibacron Blue 3GA and 4,4'-diisothyocyanostilbene-2,2'-disulphonic disodium salt selectively inhibited the Na+ currents (presumably through P2X receptors) without altering I(ATPCl), suggesting that neither P2Y nor P2X receptors are likely to be involved in I(ATPCl) activation. We conclude that I(ATPCl) is not associated with Cl- channels previously characterized in mouse parotid acinar cells, nor is it dependent on P2 nucleotide receptor stimulation. I(ATPCl) expressed in acinar cells reflects the activation of a novel ATP-gated Cl- channel that may play an important physiological role in salivary gland fluid secretion.
Figures


Comment in
-
ATP regulation of epithelial Cl- channels--new challenges?J Physiol. 2003 Feb 15;547(Pt 1):1. doi: 10.1113/jphysiol.2002.038281. Epub 2003 Jan 17. J Physiol. 2003. PMID: 12562914 Free PMC article. Review. No abstract available.
References
-
- Begenisich T, Melvin JE. Regulation of chloride channels in secretory epithelia. J Memb Biol. 1998;163:77–85. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

