Glycosylphosphatidylinositol-dependent protein trafficking in bloodstream stage Trypanosoma brucei
- PMID: 12582124
- PMCID: PMC141176
- DOI: 10.1128/EC.2.1.76-83.2003
Glycosylphosphatidylinositol-dependent protein trafficking in bloodstream stage Trypanosoma brucei
Abstract
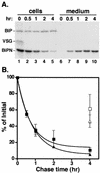
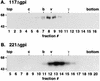
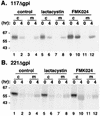
We have previously demonstrated that glycosylphosphatidylinositol (GPI) anchors strongly influence protein trafficking in the procyclic insect stage of Trypanosoma brucei (M. A. McDowell, D. A. Ransom, and J. D. Bangs, Biochem. J. 335:681-689, 1998), where GPI-minus variant surface glycoprotein (VSG) reporters have greatly reduced rates of endoplasmic reticulum (ER) exit but are ultimately secreted. We now demonstrate that GPI-dependent trafficking also occurs in pathogenic bloodstream trypanosomes. However, unlike in procyclic trypanosomes, truncated VSGs lacking C-terminal GPI-addition signals are not secreted but are mistargeted to the lysosome and degraded. Failure to export these reporters is not due to a deficiency in secretion of these cells since the N-terminal ATPase domain of the endogenous ER protein BiP is efficiently secreted from transgenic cell lines. Velocity sedimentation experiments indicate that GPI-minus VSG dimerizes similarly to wild-type VSG, suggesting that degradation is not due to ER quality control mechanisms. However, GPI-minus VSGs are fully protected from degradation by the cysteine protease inhibitor FMK024, a potent inhibitor of the major lysosomal protease trypanopain. Immunofluorescence of cells incubated with FMK024 demonstrates that GPI-minus VSG colocalizes with p67, a lysosomal marker. These data suggest that in the absence of a GPI anchor, VSG is mistargeted to the lysosome and subsequently degraded. Our findings indicate that GPI-dependent transport is a general feature of secretory trafficking in both stages of the life cycle. A working model is proposed in which GPI valence regulates progression in the secretory pathway of bloodstream stage trypanosomes.
Figures



Similar articles
-
Glycosylphosphatidylinositol-dependent secretory transport in Trypanosoma brucei.Biochem J. 1998 Nov 1;335 ( Pt 3)(Pt 3):681-9. doi: 10.1042/bj3350681. Biochem J. 1998. PMID: 9794811 Free PMC article.
-
GPI valence and the fate of secretory membrane proteins in African trypanosomes.J Cell Sci. 2005 Dec 1;118(Pt 23):5499-511. doi: 10.1242/jcs.02667. Epub 2005 Nov 15. J Cell Sci. 2005. PMID: 16291721
-
Controlling transferrin receptor trafficking with GPI-valence in bloodstream stage African trypanosomes.PLoS Pathog. 2017 May 1;13(5):e1006366. doi: 10.1371/journal.ppat.1006366. eCollection 2017 May. PLoS Pathog. 2017. PMID: 28459879 Free PMC article.
-
The glycosylation of the variant surface glycoproteins and procyclic acidic repetitive proteins of Trypanosoma brucei.Mol Biochem Parasitol. 1998 Mar 1;91(1):145-52. doi: 10.1016/s0166-6851(97)00187-4. Mol Biochem Parasitol. 1998. PMID: 9574932 Review.
-
Form and function in the trypanosomal secretory pathway.Curr Opin Microbiol. 2012 Aug;15(4):463-8. doi: 10.1016/j.mib.2012.03.002. Epub 2012 Mar 23. Curr Opin Microbiol. 2012. PMID: 22445359 Free PMC article. Review.
Cited by
-
VSG mRNA levels are regulated by the production of functional VSG protein.Mol Biochem Parasitol. 2021 Jan;241:111348. doi: 10.1016/j.molbiopara.2020.111348. Epub 2020 Dec 19. Mol Biochem Parasitol. 2021. PMID: 33352254 Free PMC article.
-
Turnover of Variant Surface Glycoprotein in Trypanosoma brucei Is Not Altered in Response to Specific Silencing.mSphere. 2022 Aug 31;7(4):e0012222. doi: 10.1128/msphere.00122-22. Epub 2022 Jun 21. mSphere. 2022. PMID: 35727016 Free PMC article.
-
Acylation in trypanosomatids: an essential process and potential drug target.Trends Parasitol. 2014 Jul;30(7):350-60. doi: 10.1016/j.pt.2014.05.003. Epub 2014 Jun 19. Trends Parasitol. 2014. PMID: 24954795 Free PMC article. Review.
-
Intracellular trafficking and glycobiology of TbPDI2, a stage-specific protein disulfide isomerase in Trypanosoma brucei.Eukaryot Cell. 2013 Jan;12(1):132-41. doi: 10.1128/EC.00293-12. Epub 2012 Nov 16. Eukaryot Cell. 2013. PMID: 23159520 Free PMC article.
-
Characterization of the late endosomal ESCRT machinery in Trypanosoma brucei.Traffic. 2013 Oct;14(10):1078-90. doi: 10.1111/tra.12094. Epub 2013 Aug 18. Traffic. 2013. PMID: 23905922 Free PMC article.
References
-
- Alexander, D., K. Schwartz, A. Balber, and J. D. Bangs. 2002. Developmentally regulated trafficking of the lysosomal membrane protein p67 in Trypanosoma brucei. J. Cell Sci. 115:3253-3263. - PubMed
-
- Bangs, J. D., E. M. Brouch, D. M. Ransom, and J. L. Roggy. 1996. A soluble secretory reporter system in Trypanosoma brucei: studies on endoplasmic reticulum targeting. J. Biol. Chem. 271:18387-18393. - PubMed
-
- Blum, M. L., J. A. Down, A. M. Gurnett, M. Carrington, M. J. Turner, and D. C. Wiley. 1993. A structural motif in the variant surface glycoprotein of Trypanosoma brucei. Nature 362:603-609. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

