Identification of a structural determinant necessary for the localization and function of estrogen receptor alpha at the plasma membrane
- PMID: 12588983
- PMCID: PMC151696
- DOI: 10.1128/MCB.23.5.1633-1646.2003
Identification of a structural determinant necessary for the localization and function of estrogen receptor alpha at the plasma membrane
Abstract
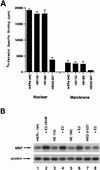
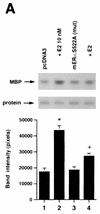
Estrogen receptors (ER) have been localized to the cell plasma membrane (PM), where signal transduction mediates some estradiol (E2) actions. However, the precise structural features of ER that result in membrane localization have not been determined. We obtained a partial tryptic peptide/mass spectrometry analysis of membrane mouse ERalpha protein. Based on this, we substituted alanine for the determined serine at amino acid 522 within the E domain of wild-type (wt) ERalpha. Upon transfection in CHO cells, the S522A mutant ERalpha resulted in a 62% decrease in membrane receptor number and reduced colocalization with caveolin 1 relative to those with expression of wt ERalpha. E2 was significantly less effective in stimulating multiple rapid signals from the membranes of CHO cells expressing ERalpha S522A than from those of CHO cells expressing wt ERalpha. In contrast, nuclear receptor expression and transcriptional function were very similar. The S522A mutant was also 60% less effective than wt ERalpha in binding caveolin 1, which facilitates ER transport to the PM. All functions of ERalpha mutants with other S-to-A substitutions were comparable to those of wt ER, and deletion of the A/B or C domain had little consequence for membrane localization or function. Transfection of ERalpha S522A into breast cancer cells that express native ER downregulated E2 binding at the membrane, signaling to ERK, and G1/S cell cycle events and progression. However, there was no effect on the E2 transactivation of an ERE-luciferase reporter. In summary, serine 522 is necessary for the efficient translocation and function of ERalpha at the PM. The S522A mutant also serves as a dominant-negative construct, identifying important functions of E2 that originate from activating PM ER.
Figures














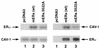



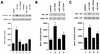




References
-
- Albanese, C., J. Johnson, G. Watanabe, N. Eklund, D. Vu, A. Arnold, and R. G. Pestell. 1995. Transforming p21 ras mutants and c-Ets-2 activate the cyclin D1 promoter through distinguishable regions. J. Biol. Chem. 270:23589-23597. - PubMed
-
- Alton, G., M. Hasilik, R. Niehues, K. Paneerselvam, J. R. Etchison, F. Fana, and H. H. Freeze. 1998. Direct utilization of mannose for mammalian glycoprotein biosynthesis. Glycobiology 8:285-295. - PubMed
-
- Anderson, R. G. 1998. The caveolae membrane system. Annu. Rev. Biochem. 67:199-225. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous
