Relaxed sugar donor selectivity of a Sinorhizobium meliloti ortholog of the Rhizobium leguminosarum mannosyl transferase LpcC. Role of the lipopolysaccharide core in symbiosis of Rhizobiaceae with plants
- PMID: 12591936
- PMCID: PMC2552401
- DOI: 10.1074/jbc.M301256200
Relaxed sugar donor selectivity of a Sinorhizobium meliloti ortholog of the Rhizobium leguminosarum mannosyl transferase LpcC. Role of the lipopolysaccharide core in symbiosis of Rhizobiaceae with plants
Abstract
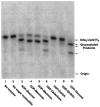
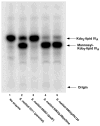
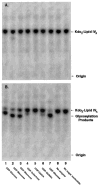
The lpcC gene of Rhizobium leguminosarum and the lpsB gene of Sinorhizobium meliloti encode protein orthologs that are 58% identical over their entire lengths of about 350 amino acid residues. LpcC and LpsB are required for symbiosis with pea and Medicago plants, respectively. S. meliloti lpsB complements a mutant of R. leguminosarum defective in lpcC, but the converse does not occur. LpcC encodes a highly selective mannosyl transferase that utilizes GDP-mannose to glycosylate the inner 3-deoxy-D-manno-octulosonic acid (Kdo) residue of the lipopolysaccharide precursor Kdo(2)-lipid IV(A). We now demonstrate that LpsB can also efficiently mannosylate the same acceptor substrate as does LpcC. Unexpectedly, however, the sugar nucleotide selectivity of LpsB is greatly relaxed compared with that of LpcC. Membranes of the wild-type S. meliloti strain 2011 catalyze the glycosylation of Kdo(2)-[4'-(32)P]lipid IV(A) at comparable rates using a diverse set of sugar nucleotides, including GDP-mannose, ADP-mannose, UDP-glucose, and ADP-glucose. This complex pattern of glycosylation is due entirely to LpsB, since membranes of the S. meliloti lpsB mutant 6963 do not glycosylate Kdo(2)-[4'-(32)P]lipid IV(A) in the presence of any of these sugar nucleotides. Expression of lpsB in E. coli using a T7lac promoter-driven construct results in the appearance of similar multiple glycosyl transferase activities seen in S. meliloti 2011 membranes. Constructs expressing lpcC display only mannosyl transferase activity. We conclude that LpsB, despite its high degree of similarity to LpcC, is a much more versatile glycosyltransferase, probably accounting for the inability of lpcC to complement S. meliloti lpsB mutants. Our findings have important implications for the regulation of core glycosylation in S. meliloti and other bacteria containing LpcC orthologs.
Figures




Similar articles
-
A mannosyl transferase required for lipopolysaccharide inner core assembly in Rhizobium leguminosarum. Purification, substrate specificity, and expression in Salmonella waaC mutants.J Biol Chem. 2003 May 2;278(18):16356-64. doi: 10.1074/jbc.M301255200. Epub 2003 Feb 17. J Biol Chem. 2003. PMID: 12591937 Free PMC article.
-
Lipopolysaccharide core glycosylation in Rhizobium leguminosarum. An unusual mannosyl transferase resembling the heptosyl transferase I of Escherichia coli.J Biol Chem. 1996 Dec 13;271(50):32119-25. J Biol Chem. 1996. PMID: 8943265
-
Genetic characterization of a Sinorhizobium meliloti chromosomal region in lipopolysaccharide biosynthesis.J Bacteriol. 2001 Feb;183(4):1248-58. doi: 10.1128/JB.183.4.1248-1258.2001. J Bacteriol. 2001. PMID: 11157937 Free PMC article.
-
Cloning and overexpression of glycosyltransferases that generate the lipopolysaccharide core of Rhizobium leguminosarum.J Biol Chem. 1998 Oct 9;273(41):26432-40. doi: 10.1074/jbc.273.41.26432. J Biol Chem. 1998. PMID: 9756877
-
Lipopolysaccharide biosynthesis in Rhizobium leguminosarum. Novel enzymes that process precursors containing 3-deoxy-D-manno-octulosonic acid.J Biol Chem. 1996 Dec 13;271(50):32112-8. J Biol Chem. 1996. PMID: 8943264
Cited by
-
Analysis of carbohydrates and glycoconjugates by matrix-assisted laser desorption/ionization mass spectrometry: An update for 2003-2004.Mass Spectrom Rev. 2009 Mar-Apr;28(2):273-361. doi: 10.1002/mas.20192. Mass Spectrom Rev. 2009. PMID: 18825656 Free PMC article. Review.
-
Expression cloning and biochemical characterization of a Rhizobium leguminosarum lipid A 1-phosphatase.J Biol Chem. 2003 Oct 10;278(41):39269-79. doi: 10.1074/jbc.M305830200. Epub 2003 Jul 16. J Biol Chem. 2003. PMID: 12869541 Free PMC article.
-
Characterization of protein glycosylation in an Asgard archaeon.BBA Adv. 2024 Jul 11;6:100118. doi: 10.1016/j.bbadva.2024.100118. eCollection 2024. BBA Adv. 2024. PMID: 39081798 Free PMC article.
-
Expression, purification, and characterization of a new Glucosyltransferase involved in the third step of O-antigen repeating-unit biosynthesis of Escherichia coli O152.Glycoconj J. 2020 Apr;37(2):139-149. doi: 10.1007/s10719-020-09907-1. Epub 2020 Jan 23. Glycoconj J. 2020. PMID: 31974821
-
A mannosyl transferase required for lipopolysaccharide inner core assembly in Rhizobium leguminosarum. Purification, substrate specificity, and expression in Salmonella waaC mutants.J Biol Chem. 2003 May 2;278(18):16356-64. doi: 10.1074/jbc.M301255200. Epub 2003 Feb 17. J Biol Chem. 2003. PMID: 12591937 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

