Infliximab or etanercept in the treatment of children with refractory juvenile idiopathic arthritis: an open label study
- PMID: 12594111
- PMCID: PMC1754468
- DOI: 10.1136/ard.62.3.245
Infliximab or etanercept in the treatment of children with refractory juvenile idiopathic arthritis: an open label study
Abstract
Objective: To study infliximab and etanercept in the treatment of refractory juvenile idiopathic arthritis (JIA).
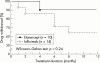
Methods: In a non-randomised, prospective, open label study, 24 patients (mean age 10.2 years, range 3.3-16.3) with polyarticular JIA were treated with either infliximab (n=14) or etanercept (n=10). The patients had had active polyarthritis for at least one year and standard treatment had failed. Anti-tumour necrosis factor (TNF) treatment was added to the current drug treatment. Infliximab (3-4 mg/kg) was given intravenously at weeks 0, 2, and 6, and thereafter at 4 to 8 week intervals. Etanercept (0.4 mg/kg) was given subcutaneously twice a week. Improvement of the patients was assessed at 3, 6, and 12 months according to established JIA response criteria.
Results: In intention to treat analyses, patients in both treatment groups improved significantly. ACR Paediatric 50 was achieved at 3, 6, and 12 months by 9/10 (90%), 8/9 (89%), and 8/9 (89%) patients with etanercept and by 8/12 (67%), 10/12 (83%), and 7/9 (78%) with infliximab, respectively. At 12 months, ACR Paediatric 75 was achieved by 67% of patients in both treatment groups. Five withdrawals due to adverse effects or lack of efficacy occurred in the infliximab group and one due to lack of compliance in the etanercept group.
Conclusion: In this open label clinical study of active JIA, both infliximab and etanercept provided a significant rapid and sustained reduction in disease activity. Adequately powered randomised controlled trials are needed to elucidate the long term safety and efficacy of TNF modulators in the treatment of JIA.
Figures

MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

