Two novel superantigens found in both group A and group C Streptococcus
- PMID: 12595453
- PMCID: PMC148831
- DOI: 10.1128/IAI.71.3.1361-1369.2003
Two novel superantigens found in both group A and group C Streptococcus
Abstract
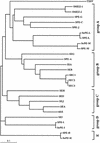
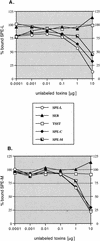
Two novel streptococcal superantigen genes (speL(Se) and speM(Se)) were identified from the Streptococcus equi genome database at the Sanger Center. Genotyping of 8 S. equi isolates and 40 Streptococcus pyogenes isolates resulted in the detection of the orthologous genes speL and speM in a restricted number of S. pyogenes isolates (15 and 5%, respectively). Surprisingly, the novel superantigen genes could not be found in any of the analyzed S. equi isolates. The results suggest that both genes are located on a mobile element that enables gene transfer between individual isolates and between streptococci from different Lancefield groups. S. equi pyrogenic exotoxin L (SPE-L(Se))/streptococcal pyrogenic exotoxin L (SPE-L) and SPE-M(Se)/SPE-M are most closely related to SMEZ, SPE-C, SPE-G, and SPE-J, but build a separate branch within this group. Recombinant SPE-L (rSPE-L) and rSPE-M were highly mitogenic for human peripheral blood lymphocytes, with half-maximum responses at 1 and 10 pg/ml, respectively. The results from competitive binding experiments suggest that both proteins bind major histocompatibility complex class II at the beta-chain, but not at the alpha-chain. The most common targets for both toxins were human Vbeta1.1 expressing T cells. Seroconversion against SPE-L and SPE-M was observed in healthy blood donors, suggesting that the toxins are expressed in vivo. Interestingly, the speL gene is highly associated with S. pyogenes M89, a serotype that is linked to acute rheumatic fever in New Zealand.
Figures


References
-
- Acharya, K. R., E. F. Passalacqua, E. Y. Jones, K. Harlos, D. I. Stuart, R. D. Brehm, and H. S. Tranter. 1994. Structural basis of superantigen action inferred from crystal structure of toxic-shock syndrome toxin-1. Nature 367:94-97. - PubMed
-
- Alouf, J. E., H. Mueller-Alouf, and W. Koehler. 1999. Superantigenic Streptococcus pyogenes erythrogenic/pyrogenic exotoxins, p. 567-588. In J. E. Alouf and J. H. Freer (ed.), Sourcebook of bacterial protein toxins. Academic Press, San Diego, Calif.
-
- Arcus, V. L., T. Proft, J. A. Sigrell, H. M. Baker, J. D. Fraser, and E. N. Baker. 2000. Conservation and variation in superantigen structure and activity highlighted by the three-dimensional structures of two new superantigens from Streptococcus pyogenes. J. Mol. Biol. 299:157-168. - PubMed
-
- Artiushin, S. C., J. F. Timoney, A. S. Sheoran, and S. K. Muthupalani. 2002. Characterization and immunogenicity of pyrogenic mitogens SePE-H and SePE-I of Streptococcus equi. Microb. Pathog. 32:71-85. - PubMed
-
- Balter, S., A. Benin, S. W. Pinto, L. M. Teixeira, G. G. Alvim, E. Luna, D. Jackson, L. LaClaire, J. Elliott, R. Facklam, and A. Schuchat. 2000. Epidemic nephritis in Nova Serrana, Brazil. Lancet 355:1776-1780. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

