Transcriptional activation by AP-2alpha is modulated by the oncogene DEK
- PMID: 12595566
- PMCID: PMC149840
- DOI: 10.1093/nar/gkg247
Transcriptional activation by AP-2alpha is modulated by the oncogene DEK
Abstract
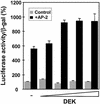
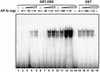
Cell differentiation and development are highly regulated processes at the transcriptional level. One of the main transcription factors that regulate these processes is AP-2alpha, a cell-type specific protein required for vertebrate development and embryogenesis. AP-2alpha also regulates apoptosis and cell-cycle specific events by interacting with the oncogene c-Myc. In searching for novel AP-2alpha- interacting factors, using an affinity chromatography approach, we have observed that oncoprotein DEK interacts with AP-2alpha in vitro. The existence of an interaction between AP-2alpha and DEK in cellular cultures was demonstrated by expression of a tagged AP-2alpha form followed by immunodetection. By transient co-expression experiments using a reporter for APOE promoter activity we have found that DEK stimulates the transactivation activity of AP-2alpha over APOE promoter. Finally, electrophoretic mobility shift assays suggested that DEK enhances the DNA-binding activity of AP-2alpha. Our data suggest a novel cellular function of DEK as a transcriptional co-activator.
Figures


Similar articles
-
Regulation of DEK expression by AP-2α and methylation level of DEK promoter in hepatocellular carcinoma.Oncol Rep. 2016 Oct;36(4):2382-90. doi: 10.3892/or.2016.4984. Epub 2016 Jul 28. Oncol Rep. 2016. PMID: 27499261
-
Functional characterization of the interacting domains of the positive coactivator PC4 with the transcription factor AP-2alpha.Gene. 2003 Nov 27;320:155-64. doi: 10.1016/s0378-1119(03)00823-0. Gene. 2003. PMID: 14597399
-
Dual regulation of AP-2alpha transcriptional activation by poly(ADP-ribose) polymerase-1.Biochem J. 2004 Aug 15;382(Pt 1):323-9. doi: 10.1042/BJ20040593. Biochem J. 2004. PMID: 15170387 Free PMC article.
-
The Complexity of DEK Signaling in Cancer Progression.Curr Cancer Drug Targets. 2018;18(3):256-265. doi: 10.2174/1568009617666170522094730. Curr Cancer Drug Targets. 2018. PMID: 28530531 Review.
-
Role of the DEK oncogene in the development of squamous cell carcinoma.Int J Clin Oncol. 2020 Sep;25(9):1563-1569. doi: 10.1007/s10147-020-01735-5. Epub 2020 Jul 12. Int J Clin Oncol. 2020. PMID: 32656741 Free PMC article. Review.
Cited by
-
Novel molecular mechanisms in Alzheimer's disease: The potential role of DEK in disease pathogenesis.Front Aging Neurosci. 2022 Oct 6;14:1018180. doi: 10.3389/fnagi.2022.1018180. eCollection 2022. Front Aging Neurosci. 2022. PMID: 36275000 Free PMC article. Review.
-
DEK Expression in Breast Cancer Cells Leads to the Alternative Activation of Tumor Associated Macrophages.Cancers (Basel). 2020 Jul 17;12(7):1936. doi: 10.3390/cancers12071936. Cancers (Basel). 2020. PMID: 32708944 Free PMC article.
-
DEK proto-oncogene expression interferes with the normal epithelial differentiation program.Am J Pathol. 2009 Jan;174(1):71-81. doi: 10.2353/ajpath.2009.080330. Epub 2008 Nov 26. Am J Pathol. 2009. PMID: 19036808 Free PMC article.
-
Stacking the DEK: from chromatin topology to cancer stem cells.Cell Cycle. 2013 Jan 1;12(1):51-66. doi: 10.4161/cc.23121. Epub 2012 Dec 19. Cell Cycle. 2013. PMID: 23255114 Free PMC article. Review.
-
Dysregulated Transcription Factor TFAP2A After Peripheral Nerve Injury Modulated Schwann Cell Phenotype.Neurochem Res. 2019 Dec;44(12):2776-2785. doi: 10.1007/s11064-019-02898-y. Epub 2019 Oct 25. Neurochem Res. 2019. PMID: 31650361
References
-
- Williams T., Admon,A., Luscher,B. and Tjian,R. (1988) Cloning and expression of AP-2, a cell-type-specific transcription factor that activates inducible enhancer elements. Genes Dev., 2, 1557–1569. - PubMed
-
- Mitchell P.J., Wang,C. and Tjian,R. (1987) Positive and negative regulation of transcription in vitro: enhancer-binding protein AP-2 is inhibited by SV40 T antigen. Cell, 50, 847–861. - PubMed
-
- Moser M., Imhof,A., Pscherer,A., Bauer,R., Amselgruber,W., Sinowatz,F., Hofstadter,F., Schule,R. and Buettner,R. (1995) Cloning and characterization of a second AP-2 transcription factor: AP-2β. Development, 121, 2779–2788. - PubMed
-
- Oulad-Abdelghani M., Bouillet,P., Chazaud,C., Dolle,P. and Chambon,P. (1996) AP-2.2: a novel AP-2-related transcription factor induced by retinoic acid during differentiation of P19 embryonal carcinoma cells. Exp. Cell Res., 225, 338–347. - PubMed
-
- Zhao F., Satoda,M., Licht,J.D., Hayashizaki,Y. and Gelb,B.D. (2001) Cloning and characterization of a novel mouse AP-2 transcription factor, AP-2δ, with unique DNA binding and transactivation properties. J. Biol. Chem., 276, 40755–40760. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

