Transcription profiling of platelet-derived growth factor-B-deficient mouse embryos identifies RGS5 as a novel marker for pericytes and vascular smooth muscle cells
- PMID: 12598306
- PMCID: PMC1868109
- DOI: 10.1016/S0002-9440(10)63868-0
Transcription profiling of platelet-derived growth factor-B-deficient mouse embryos identifies RGS5 as a novel marker for pericytes and vascular smooth muscle cells
Abstract
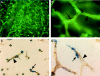
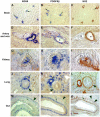
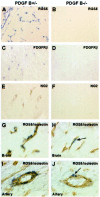
All blood capillaries consist of endothelial tubes surrounded by mural cells referred to as pericytes. The origin, recruitment, and function of the pericytes is poorly understood, but the importance of these cells is underscored by the severe cardiovascular defects in mice genetically devoid of factors regulating pericyte recruitment to embryonic vessels, and by the association between pericyte loss and microangiopathy in diabetes mellitus. A general problem in the study of pericytes is the shortage of markers for these cells. To identify new markers for pericytes, we have taken advantage of the platelet-derived growth factor (PDGF)-B knockout mouse model, in which developing blood vessels in the central nervous system are almost completely devoid of pericytes. Using cDNA microarrays, we analyzed the gene expression in PDGF-B null embryos in comparison with corresponding wild-type embryos and searched for down-regulated genes. The most down-regulated gene present on our microarray was RGS5, a member of the RGS family of GTPase-activating proteins for G proteins. In situ hybridization identified RGS5 expression in brain pericytes, and in pericytes and vascular smooth muscle cells in certain other, but not all, locations. Absence of RGS5 expression in PDGF-B and PDGFR beta-null embryos correlated with pericyte loss in these mice. Residual RGS5 expression in rare pericytes suggested that RGS5 is a pericyte marker expressed independently of PDGF-B/R beta signaling. With RGS5 as a proof-of-principle, our data demonstrate the usefulness of microarray analysis of mouse models for abnormal pericyte development in the identification of new pericyte-specific markers.
Figures
References
-
- Allt G, Lawrenson JG: Pericytes: cell biology and pathology. Cells Tissues Organs 2001, 169:1-11 - PubMed
-
- Sims DE: Diversity within pericytes. Clin Exp Pharmacol Physiol 2000, 27:842-846 - PubMed
-
- Dickson MC, Martin JS, Cousins FM, Kulkarni AB, Karlsson S, Akhurst RJ: Defective haematopoiesis and vasculogenesis in transforming growth factor beta-1 knock-out mice. Development 1995, 121:1845-1854 - PubMed
-
- Li DY, Sorensen LK, Brooke BS, Urness LD, Davis EC, Taylor DG, Boak BB, Wendel DP: Defective angiogenesis in mice lacking endoglin. Science 1999, 284:1534-1537 - PubMed
-
- Yang X, Castilla LH, Xu X, Li C, Gotay J, Weinstein M, Liu PP, Deng CX: Angiogenesis defects and mesenchymal apoptosis in mice lacking SMAD5. Development 1999, 126:1571-1580 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

