TACE is required for the activation of the EGFR by TGF-alpha in tumors
- PMID: 12606576
- PMCID: PMC150344
- DOI: 10.1093/emboj/cdg111
TACE is required for the activation of the EGFR by TGF-alpha in tumors
Abstract
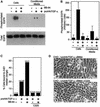
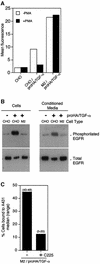
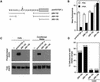
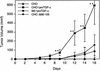
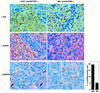
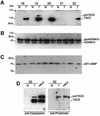
The factors and mechanisms that transduce the intracellular signals sent upon activation of the receptor for the epidermal growth factor (EGFR) and related receptors are reasonably well understood and, in fact, are the targets of anti-tumor drugs. In contrast, less is known about the mechanisms implicated in sending the signals that activate these receptors. Here we show that when its proteolytic shedding is prevented, the transmembrane form of the transforming growth factor-alpha (proTGF-alpha) interacts with, but does not activate, the EGFR. Thus, shedding seems to control not only the availability of the soluble form of the growth factor (TGF-alpha) but also the activity of the transmembrane form. The activity of the protease responsible for the shedding of proTGF-alpha, tumor necrosis factor-alpha converting enzyme (TACE), is required for the activation of the EGFR in vivo and for the development of tumors in nude mice, indicating a crucial role of TACE in tumorigenesis. In agreement with this view, TACE is dramatically overexpressed in the majority of mammary tumors analyzed. Collectively, this evidence points to TACE as a promising target of anti-tumor therapy.
Figures

References
-
- Albanell J. et al. (2001) Activated extracellular signal-regulated kinases: association with epidermal growth factor receptor/transforming growth factor-α expression in head and neck squamous carcinoma and inhibition by anti-epidermal growth factor receptor treatments. Cancer Res., 61, 6500–6510. - PubMed
-
- Albanell J. et al. (2002) Pharmacodynamic studies of the epidermal growth factor receptor inhibitor ZD1839 in skin from cancer patients: histopathologic and molecular consequences of receptor inhibition. J. Clin. Oncol., 20, 110–124. - PubMed
-
- Arribas J. and Merlos-Suárez,A. (2002) Shedding of plasma membrane proteins. Curr. Top. Dev. Biol., in press. - PubMed
-
- Arribas J., Coodly,L., Vollmer,P., Kishimoto,T.K., Rosejohn,S. and Massagué,J. (1996) Diverse cell surface protein ectodomains are shed by a system sensitive to metalloprotease inhibitors. J. Biol. Chem., 271, 11376–11382. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

