Human immunodeficiency virus type 1 infection and replication in normal human oral keratinocytes
- PMID: 12610122
- PMCID: PMC149546
- DOI: 10.1128/jvi.77.6.3470-3476.2003
Human immunodeficiency virus type 1 infection and replication in normal human oral keratinocytes
Abstract
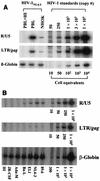
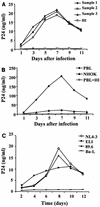
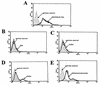
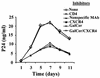
Recent epidemiologic studies show increasing human immunodeficiency virus type 1 (HIV-1) transmission through oral-genital contact. This paper examines the possibility that normal human oral keratinocytes (NHOKs) might be directly infected by HIV or might convey infectious HIV virions to adjacent leukocytes. PCR analysis of proviral DNA constructs showed that NHOKs can be infected by CXCR4-tropic (NL4-3 and ELI) and dualtropic (89.6) strains of HIV-1 to generate a weak but productive infection. CCR5-tropic strain Ba-L sustained minimal viral replication. Antibody inhibition studies showed that infection by CXCR4-tropic viral strains is mediated by the galactosylceramide receptor and the CXCR4 chemokine coreceptor. Coculture studies showed that infectious HIV-1 virions can also be conveyed from NHOKs to activated peripheral blood lymphocytes, suggesting a potential role of oral epithelial cells in the transmission of HIV infection.
Figures
Similar articles
-
Oral keratinocytes support non-replicative infection and transfer of harbored HIV-1 to permissive cells.Retrovirology. 2008 Jul 17;5:66. doi: 10.1186/1742-4690-5-66. Retrovirology. 2008. PMID: 18637194 Free PMC article.
-
Oral epithelial cells are susceptible to cell-free and cell-associated HIV-1 infection in vitro.Virology. 2003 Sep 1;313(2):343-53. doi: 10.1016/s0042-6822(03)00283-6. Virology. 2003. PMID: 12954203
-
Alcohol enhances HIV type 1 infection in normal human oral keratinocytes by up-regulating cell-surface CXCR4 coreceptor.AIDS Res Hum Retroviruses. 2004 May;20(5):513-9. doi: 10.1089/088922204323087769. AIDS Res Hum Retroviruses. 2004. PMID: 15186526
-
The effects of HIV infection on oral mucosal immunity.Adv Dent Res. 2006 Apr 1;19(1):29-35. doi: 10.1177/154407370601900107. Adv Dent Res. 2006. PMID: 16672546 Review.
-
Plausibility of HIV-1 Infection of Oral Mucosal Epithelial Cells.Adv Dent Res. 2011 Apr;23(1):38-44. doi: 10.1177/0022034511399283. Adv Dent Res. 2011. PMID: 21441479 Free PMC article. Review.
Cited by
-
Oral and vaginal epithelial cell lines bind and transfer cell-free infectious HIV-1 to permissive cells but are not productively infected.PLoS One. 2014 May 23;9(5):e98077. doi: 10.1371/journal.pone.0098077. eCollection 2014. PLoS One. 2014. PMID: 24857971 Free PMC article.
-
The mouth: a gateway or a trap for HIV?AIDS. 2010 Jan 2;24(1):5-16. doi: 10.1097/QAD.0b013e328333525f. AIDS. 2010. PMID: 19935380 Free PMC article. Review. No abstract available.
-
Involvement of claudin-7 in HIV infection of CD4(-) cells.Retrovirology. 2005 Dec 20;2:79. doi: 10.1186/1742-4690-2-79. Retrovirology. 2005. PMID: 16368003 Free PMC article.
-
Visualization of X4- and R5-Tropic HIV-1 Viruses Expressing Fluorescent Proteins in Human Endometrial Cells: Application to Tropism Study.PLoS One. 2017 Jan 6;12(1):e0169453. doi: 10.1371/journal.pone.0169453. eCollection 2017. PLoS One. 2017. PMID: 28060897 Free PMC article.
-
Transcytosis as a Mechanism of HIV-1 Entry into Columnar Epithelial Explants of the Female Reproductive Tract.AIDS Res Hum Retroviruses. 2025 Mar;41(3):167-173. doi: 10.1089/aid.2024.0045. Epub 2024 Dec 12. AIDS Res Hum Retroviruses. 2025. PMID: 39665594
References
-
- Alfsen, A., and M. Bomsel. 2002. HIV-1 gp41 envelope residues 650-685 exposed on native virus act as a lectin to bind epithelial cell galactosyl ceramide. J. Biol. Chem. 12:25649-25659. - PubMed
-
- Baba, T. W., A. M. Trichel, L. An, V. Liska, L. N. Martin, M. Murphey-Corb, and R. M. Ruprecht. 1996. Infection and AIDS in adult macaques after nontraumatic oral exposure to cell-free SIV. Science 272:1486-1489. - PubMed
-
- Bergey, E. J., M. I. Cho, B. M. Blumberg, M. L. Hammarskjold, D. Rekosh, L. G. Epstein, and M. J. Levine. 1994. Interaction of HIV-1 and human salivary mucins. J. Acquir. Immune Defic. Syndr. 7:995-1002. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

