Role of integrin-linked kinase in nerve growth factor-stimulated neurite outgrowth
- PMID: 12629168
- PMCID: PMC6741947
- DOI: 10.1523/JNEUROSCI.23-05-01638.2003
Role of integrin-linked kinase in nerve growth factor-stimulated neurite outgrowth
Abstract
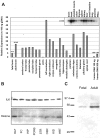
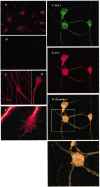
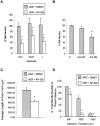
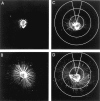
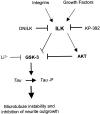
The role of integrin-linked kinase (ILK), a kinase that is involved in various cellular processes, including adhesion and migration, has not been studied in primary neurons. Using mRNA dot blot and Western blot analysis of ILK in rat and human brain tissue, we found that ILK is expressed in various regions of the CNS. Immunohistochemical and immunocytochemical techniques revealed granular ILK staining that is enriched in neurons and colocalizes with the beta1 integrin subunit. The role of ILK in neurite growth promotion by NGF was studied in rat pheochromocytoma cells and dorsal root ganglion neurons using a pharmacological inhibitor of ILK (KP-392) or after overexpression of dominant-negative ILK (ILK-DN). Both molecular and pharmacological inhibition of ILK activity significantly reduced NGF-induced neurite outgrowth. Survival assays indicate that KP-392-induced suppression of neurite outgrowth occurred in the absence of cell death. ILK kinase activity was stimulated by NGF. NGF-mediated stimulation of phosphorylation of both AKT and the Tau kinase glycogen synthase kinase-3 (GSK-3) was inhibited in the presence of KP-392 and after overexpression of ILK-DN. Consequently, ILK inhibition resulted in an increase in the hyperphosphorylation of Tau, a substrate of GSK-3. Together these findings indicate that ILK is an important effector in NGF-mediated neurite outgrowth.
Figures





Similar articles
-
Inactivation of integrin-linked kinase induces aberrant tau phosphorylation via sustained activation of glycogen synthase kinase 3beta in N1E-115 neuroblastoma cells.J Biol Chem. 2003 Jul 18;278(29):26970-5. doi: 10.1074/jbc.M304113200. Epub 2003 Apr 24. J Biol Chem. 2003. PMID: 12714590
-
Integrin activation and neurotrophin signaling cooperate to enhance neurite outgrowth in sensory neurons.J Comp Neurol. 2005 Jun 6;486(3):267-80. doi: 10.1002/cne.20518. J Comp Neurol. 2005. PMID: 15844170
-
The synergistic effects of NGF and IGF-1 on neurite growth in adult sensory neurons: convergence on the PI 3-kinase signaling pathway.J Neurochem. 2003 Sep;86(5):1116-28. doi: 10.1046/j.1471-4159.2003.01925.x. J Neurochem. 2003. PMID: 12911620
-
Integrin-linked kinase (ILK): a "hot" therapeutic target.Biochem Pharmacol. 2000 Oct 15;60(8):1115-9. doi: 10.1016/s0006-2952(00)00444-5. Biochem Pharmacol. 2000. PMID: 11007949 Review.
-
The role of integrin-linked kinase (ILK) in cancer progression.Cancer Metastasis Rev. 2003 Dec;22(4):375-84. doi: 10.1023/a:1023777013659. Cancer Metastasis Rev. 2003. PMID: 12884912 Review.
Cited by
-
Concurrent nicotine exposure to prenatal alcohol consumption alters the hippocampal and cortical neurotoxicity.Heliyon. 2020 Jan 8;6(1):e03045. doi: 10.1016/j.heliyon.2019.e03045. eCollection 2020 Jan. Heliyon. 2020. PMID: 31938742 Free PMC article.
-
The inositol polyphosphate 5-phosphatase, PIPP, Is a novel regulator of phosphoinositide 3-kinase-dependent neurite elongation.Mol Biol Cell. 2006 Feb;17(2):607-22. doi: 10.1091/mbc.e05-05-0469. Epub 2005 Nov 9. Mol Biol Cell. 2006. PMID: 16280363 Free PMC article.
-
Regulation of mitotic cytoskeleton dynamics and cytokinesis by integrin-linked kinase in retinoblastoma cells.PLoS One. 2014 Jun 9;9(6):e98838. doi: 10.1371/journal.pone.0098838. eCollection 2014. PLoS One. 2014. PMID: 24911651 Free PMC article.
-
Activation of casein kinase II and inhibition of phosphatase and tensin homologue deleted on chromosome 10 phosphatase by nerve growth factor/p75NTR inhibit glycogen synthase kinase-3beta and stimulate axonal growth.Mol Biol Cell. 2006 Aug;17(8):3369-77. doi: 10.1091/mbc.e05-12-1144. Epub 2006 May 24. Mol Biol Cell. 2006. PMID: 16723502 Free PMC article.
-
Integrin-linked kinase mediates bone morphogenetic protein 7-dependent renal epithelial cell morphogenesis.Mol Cell Biol. 2005 May;25(9):3648-57. doi: 10.1128/MCB.25.9.3648-3657.2005. Mol Cell Biol. 2005. PMID: 15831470 Free PMC article.
References
-
- Andjelkovic M, Alessi DR, Meier R, Fernandez A, Lamb NJ, Frech M, Cron M, Cohen P, Lucocq JM, Hemmings BA. Role of translocation in the activation and function of protein kinase B. J Biol Chem. 1997;272:31515–31524. - PubMed
-
- Andjelkovic M, Suidan HS, Meier R, Frech M, Alessi DR, Hemmings BA. Nerve growth factor promotes activation of the α, β and γ isoforms of protein kinase B in PC12 pheochromocytoma cells. Eur J Biochem. 1998;251:195–200. - PubMed
-
- Ashcroft M, Stephens RM, Hallberg B, Downward J, Kaplan DR. The selective and inducible activation of endogenous PI 3-kinase in PC12 cells results in efficient NGF-mediated survival but defective neurite outgrowth. Oncogene. 1999;18:4586–4597. - PubMed
-
- Attwell S, Roskelley C, Dedhar S. The integrin-linked kinase (ILK) suppresses anoikis. Oncogene. 2000;19:3811–3815. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous
