Reversible disorganization of the locomotor pattern after neonatal spinal cord transection in the rat
- PMID: 12629197
- PMCID: PMC6741960
- DOI: 10.1523/JNEUROSCI.23-05-01924.2003
Reversible disorganization of the locomotor pattern after neonatal spinal cord transection in the rat
Abstract
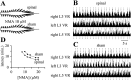
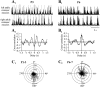
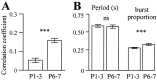
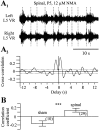
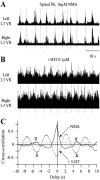
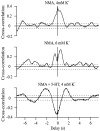
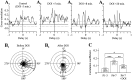
The central pattern generators (CPGs) for locomotion, located in the lumbar spinal cord, are functional at birth in the rat. Their maturation occurs during the last few days preceding birth, a period during which the first projections from the brainstem start to reach the lumbar enlargement of the spinal cord. The goal of the present study was to investigate the effect of suppressing inputs from supraspinal structures on the CPGs, shortly after their formation. The spinal cord was transected at the thoracic level at birth [postnatal day 0 (P0)]. We examined during the first postnatal week the capacity of the CPGs to produce rhythmic motor activity in two complementary experimental conditions. Left and right ankle extensor muscles were recorded in vivo during airstepping, and lumbar ventral roots were recorded in vitro during pharmacologically evoked fictive locomotion. Mechanical stimulation of the tail elicited long-lasting sequences of airstepping in the spinal neonates and only a few steps in sham-operated rats. In vitro experiments made simultaneously on spinal and sham animals confirmed the increased excitability of the CPGs after spinalization. A left-right alternating locomotor pattern was observed at P1-P3. Both types of experiments showed that the pattern was disorganized at P6-P7, and that the left-right alternation was lost. Alternation was restored after the activation of serotonergic 5-HT(2) receptors in vivo. These results suggest that descending pathways, in particular serotonergic projections, control the strength of reciprocal inhibition and therefore shape the locomotor pattern in the neonatal rat.
Figures

References
-
- Andén N-E, Häggendal J, Magnusson T, Rosengren E. The time course of the disappearance of noradrenaline and 5-hydroxytryptamine in the spinal cord after transection. Acta Physiol Scand. 1964;62:115–118. - PubMed
-
- Barbeau H, Rossignol S. Recovery of locomotion after chronic spinalization in the adult cat. Brain Res. 1987;412:84–95. - PubMed
-
- Barbeau H, Rossignol S. The effects of serotonergic drugs on the locomotor pattern and on cutaneous reflexes of the adult chronic spinal cat. Brain Res. 1990;514:55–67. - PubMed
-
- Bradley NS, Smith JL. Neuromuscular patterns of stereotypic hindlimb behaviors in the first two postnatal months. II. Stepping in spinal kittens. Brain Res. 1988;466:53–67. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
