Role of CD31/platelet endothelial cell adhesion molecule-1 expression in in vitro and in vivo growth and differentiation of human breast cancer cells
- PMID: 12651608
- PMCID: PMC1851235
- DOI: 10.1016/S0002-9440(10)63912-0
Role of CD31/platelet endothelial cell adhesion molecule-1 expression in in vitro and in vivo growth and differentiation of human breast cancer cells
Abstract
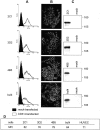
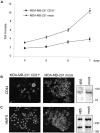
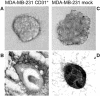
Breast ductal carcinoma in situ is an intraductal proliferation of malignant epithelial cells that diffuse within the ductal system without stromal invasion. Our finding that a subset of these tumors express CD31/platelet endothelial cell adhesion molecule-1 suggests that breast cancer represents an informative model for studying the involvement of the molecule in the morphogenesis, differentiation, and diffusion of this disease. Transfection of CD31 in MDA-MB-231 cells caused reduction in growth, loss of CD44, and acquisition of a ductal morphology. The same effects were maintained in vivo, in which CD31(+) tumors grew with in situ-like aspects, papillary differentiation, and a secretory phenotype. CD44 was down-modulated, with the CD31(+) cells blocked in the G(1) phase. The morphology was highly similar to what was observed in some human CD31(+) ductal carcinomas in situ. MDA-MB-231 mock cells grew in solid sheets, lacking stromal material, and displaying high levels of CD44 and proliferation. CD31(+) cells acquired motility characteristics in in vitro assays, a finding confirmed in vivo by the diffusion of human tumor cells throughout the normal ducts residual in the murine mammary gland. In conclusion, CD31 expression reverts the undifferentiated morphology and aggressive behavior of MDA-MB-231 cells, indicating its active role in the morphogenesis of breast ductal in situ carcinomas.
Figures





Similar articles
-
Expression of CD31 by cells of extensive ductal in situ and invasive carcinomas of the breast.J Pathol. 2001 Jun;194(2):254-61. doi: 10.1002/1096-9896(200106)194:2<254::AID-PATH880>3.0.CO;2-2. J Pathol. 2001. PMID: 11400156
-
Monitoring metastatic behavior of human tumor cells in mice with species-specific polymerase chain reaction: elevated expression of angiogenesis and bone resorption stimulators by breast cancer in bone metastases.J Bone Miner Res. 2001 Jun;16(6):1077-91. doi: 10.1359/jbmr.2001.16.6.1077. J Bone Miner Res. 2001. PMID: 11393785
-
Vascular endothelial growth inhibitor affects the invasion, apoptosis and vascularisation in breast cancer cell line MDA-MB-231.Chin Med J (Engl). 2014;127(10):1947-53. Chin Med J (Engl). 2014. PMID: 24824261
-
Biologic role of activated leukocyte cell adhesion molecule overexpression in breast cancer cell lines and clinical tumor tissue.Breast Cancer Res Treat. 2011 Sep;129(2):347-60. doi: 10.1007/s10549-010-1219-y. Epub 2010 Oct 23. Breast Cancer Res Treat. 2011. PMID: 20972617
-
ALCAM, activated leukocyte cell adhesion molecule, influences the aggressive nature of breast cancer cells, a potential connection to bone metastasis.Anticancer Res. 2010 Apr;30(4):1163-8. Anticancer Res. 2010. PMID: 20530423
Cited by
-
Low-Magnitude High-Frequency Vibration Accelerated the Foot Wound Healing of n5-streptozotocin-induced Diabetic Rats by Enhancing Glucose Transporter 4 and Blood Microcirculation.Sci Rep. 2017 Sep 14;7(1):11631. doi: 10.1038/s41598-017-11934-2. Sci Rep. 2017. PMID: 28912573 Free PMC article.
-
Cell aggregation induces phosphorylation of PECAM-1 and Pyk2 and promotes tumor cell anchorage-independent growth.Mol Cancer. 2010 Jan 14;9:7. doi: 10.1186/1476-4598-9-7. Mol Cancer. 2010. PMID: 20074345 Free PMC article.
-
Exploring the elasticity and adhesion behavior of cardiac fibroblasts by atomic force microscopy indentation.Mater Sci Eng C Mater Biol Appl. 2014 Jul 1;40:427-34. doi: 10.1016/j.msec.2014.04.003. Epub 2014 Apr 13. Mater Sci Eng C Mater Biol Appl. 2014. PMID: 24857511 Free PMC article.
-
Application of bioactive materials primary cilia as a novel delivery vehicle.Bioact Mater. 2025 May 8;51:70-82. doi: 10.1016/j.bioactmat.2025.04.015. eCollection 2025 Sep. Bioact Mater. 2025. PMID: 40463394 Free PMC article.
-
The prognostic value of platelet endothelial cell adhesion molecule-1 in non-small-cell lung cancer patients.Med Oncol. 2013 Jun;30(2):536. doi: 10.1007/s12032-013-0536-5. Epub 2013 Mar 15. Med Oncol. 2013. PMID: 23494670
References
-
- DeLisser HM, Newman PJ, Albelda SM: Molecular and functional aspects of PECAM-1/CD31. Immunol Today 1994, 15:490-495 - PubMed
-
- Muller WA, Randolph GJ: Migration of leukocytes across endothelium and beyond: molecules involved in the transmigration and fate of monocytes. J Leukoc Biol 1999, 66:698-704 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

