Use of genetically engineered phage to deliver antimicrobial agents to bacteria: an alternative therapy for treatment of bacterial infections
- PMID: 12654662
- PMCID: PMC152521
- DOI: 10.1128/AAC.47.4.1301-1307.2003
Use of genetically engineered phage to deliver antimicrobial agents to bacteria: an alternative therapy for treatment of bacterial infections
Abstract
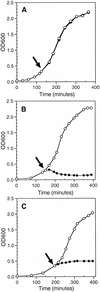
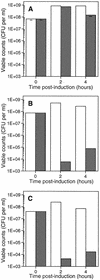
The emergence and increasing prevalence of multidrug-resistant bacterial pathogens emphasizes the need for new and innovative antimicrobial strategies. Lytic phages, which kill their host following amplification and release of progeny phage into the environment, may offer an alternative strategy for combating bacterial infections. In this study, however, we describe the use of a nonlytic phage to specifically target and deliver DNA encoding bactericidal proteins to bacteria. To test the concept of using phage as a lethal-agent delivery vehicle, we used the M13 phagemid system and the addiction toxins Gef and ChpBK. Phage delivery of lethal-agent phagemids reduced target bacterial numbers by several orders of magnitude in vitro and in a bacteremic mouse model of infection. Given the powerful genetic engineering tools available and the present knowledge in phage biology, this technology may have potential use in antimicrobial therapies and DNA vaccine development.
Figures



References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

