Oral mucositis and selective elimination of oral flora in head and neck cancer patients receiving radiotherapy: a double-blind randomised clinical trial
- PMID: 12671696
- PMCID: PMC2376383
- DOI: 10.1038/sj.bjc.6600824
Oral mucositis and selective elimination of oral flora in head and neck cancer patients receiving radiotherapy: a double-blind randomised clinical trial
Abstract
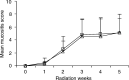
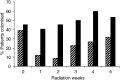
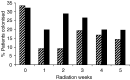
Mucositis is an acute inflammation of the oral mucosa because of radiotherapy and/or chemotherapy. All patients receiving radiotherapy in the head and neck region develop oral mucositis. The aim of this study was to analyse the effects of selective oral flora elimination on radiotherapy-induced oral mucositis, in a double-blind, randomised, placebo-controlled trial. Sixty-five patients with a malignant tumour in the head and neck regions to be treated with primary curative or postoperative radiotherapy participated in this study. The patients received either the active lozenges of 1 g containing polymyxin E 2 mg, tobramycin 1.8 mg and amphotericin B 10 mg (PTA) (33 patients) or the placebo lozenges (32 patients), four times daily during the full course of radiotherapy. Mucositis, changes in the oral flora, quality of feeding and changes of total body weight were assessed. Mucositis score did not differ between the groups during the first 5 weeks of radiotherapy. Nasogastric tube feeding was needed in six patients (19%) of the placebo group and two patients (6%) of the PTA group (P=0.08). Mean weight loss after 5 weeks of radiation was less in the PTA group (1.3 kg) (s.d.: 3.0) than in the placebo group (2.8 kg) (s.d.: 2.9) (P=0.05). Colonisation index of Candida species and Gram-negative bacilli was reduced in the PTA group and not in the placebo group (P<0.05). No effect on other microorganisms was detected. In conclusion, selective oral flora elimination in head and neck irradiation patients does not prevent the development of severe mucositis.
Figures
References
-
- Anonymous (1979) Handbook for Reporting Results of Cancer Treatment, Vol. 48. 15–22. WHO Offset Publication.
-
- Anonymous (1993) ICRU Report 50: Prescribing, Recording and Reporting Photon Beam Therapy. Bethesda, MD: ICRU Publications
-
- Baker DG. (1982) The radiobiological basis for tissue reactions in the oral cavity following therapeutic X-irradiation. A review. Arch Otolaryngol 108: 21–24 - PubMed
-
- Bernhoft CH, Skaug N (1985) Oral findings in irradiated edentulous patients. Int J Oral Surg 14: 416–427 - PubMed
-
- Jansma J, Vissink A, Spijkervet FKL, Roodenburg JLN, Panders AK, Vermey A, Szabo BG, Gravenmade EJ (1992) Protocol for the prevention and treatment of oral sequelae resulting from head and neck radiation therapy. Cancer 70: 2171–2180 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

