Heat shock response by the hyperthermophilic archaeon Pyrococcus furiosus
- PMID: 12676722
- PMCID: PMC154833
- DOI: 10.1128/AEM.69.4.2365-2371.2003
Heat shock response by the hyperthermophilic archaeon Pyrococcus furiosus
Abstract
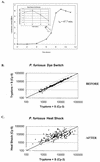
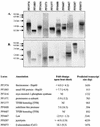
Collective transcriptional analysis of heat shock response in the hyperthermophilic archaeon Pyrococcus furiosus was examined by using a targeted cDNA microarray in conjunction with Northern analyses. Differential gene expression suggests that P. furiosus relies on a cooperative strategy of rescue (thermosome [Hsp60], small heat shock protein [Hsp20], and two VAT-related chaperones), proteolysis (proteasome), and stabilization (compatible solute formation) to cope with polypeptide processing during thermal stress.
Figures

Similar articles
-
Role of the beta1 subunit in the function and stability of the 20S proteasome in the hyperthermophilic archaeon Pyrococcus furiosus.J Bacteriol. 2007 Jan;189(2):583-90. doi: 10.1128/JB.01382-06. Epub 2006 Nov 17. J Bacteriol. 2007. PMID: 17114253 Free PMC article.
-
Microarray analysis of the hyperthermophilic archaeon Pyrococcus furiosus exposed to gamma irradiation.Extremophiles. 2007 Jan;11(1):19-29. doi: 10.1007/s00792-006-0002-9. Epub 2006 Aug 8. Extremophiles. 2007. PMID: 16896524
-
A novel archaeal transcriptional regulator of heat shock response.J Biol Chem. 2003 Jan 3;278(1):18-26. doi: 10.1074/jbc.M209250200. Epub 2002 Oct 14. J Biol Chem. 2003. PMID: 12381724
-
The role of TrmB and TrmB-like transcriptional regulators for sugar transport and metabolism in the hyperthermophilic archaeon Pyrococcus furiosus.Arch Microbiol. 2008 Sep;190(3):247-56. doi: 10.1007/s00203-008-0378-2. Epub 2008 May 11. Arch Microbiol. 2008. PMID: 18470695 Review.
-
The Development of Tungsten Biochemistry-A Personal Recollection.Molecules. 2023 May 11;28(10):4017. doi: 10.3390/molecules28104017. Molecules. 2023. PMID: 37241758 Free PMC article. Review.
Cited by
-
Role of the beta1 subunit in the function and stability of the 20S proteasome in the hyperthermophilic archaeon Pyrococcus furiosus.J Bacteriol. 2007 Jan;189(2):583-90. doi: 10.1128/JB.01382-06. Epub 2006 Nov 17. J Bacteriol. 2007. PMID: 17114253 Free PMC article.
-
Transcriptional and biochemical analysis of starch metabolism in the hyperthermophilic archaeon Pyrococcus furiosus.J Bacteriol. 2006 Mar;188(6):2115-25. doi: 10.1128/JB.188.6.2115-2125.2006. J Bacteriol. 2006. PMID: 16513741 Free PMC article.
-
Impact of substrate glycoside linkage and elemental sulfur on bioenergetics of and hydrogen production by the hyperthermophilic archaeon Pyrococcus furiosus.Appl Environ Microbiol. 2007 Nov;73(21):6842-53. doi: 10.1128/AEM.00597-07. Epub 2007 Sep 7. Appl Environ Microbiol. 2007. PMID: 17827328 Free PMC article.
-
Draft genome sequence of sulfur-reducing archaeon Thermococcus thioreducens DSM 14981T.Braz J Microbiol. 2017 Jan-Mar;48(1):3-4. doi: 10.1016/j.bjm.2016.04.031. Epub 2016 Jun 4. Braz J Microbiol. 2017. PMID: 27328615 Free PMC article.
-
Dynamic metabolic adjustments and genome plasticity are implicated in the heat shock response of the extremely thermoacidophilic archaeon Sulfolobus solfataricus.J Bacteriol. 2006 Jun;188(12):4553-9. doi: 10.1128/JB.00080-06. J Bacteriol. 2006. PMID: 16740961 Free PMC article.
References
-
- Alexandre, H., V. Ansanay-Galeote, S. Dequin, and B. Blondin. 2001. Global gene expression during short term ethanol stress in Saccharomyces cerevisiae. FEBS Lett. 498:98-103. - PubMed
-
- Bell, S. D., and S. P. Jackson. 1998. Transcription and translation in archaea: a mosaic of eukaryl and bacterial features. Trends Microbiol. 6:222-228. - PubMed
-
- Bouyoub, A., G. Barbier, J. Querellou, and P. Forterre. 1995. A putative SOS repair gene (dinF-like) in a hyperthermophilic archaeon. Gene 167:147-149. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

