Evidence that the retroviral DNA integration process triggers an ATR-dependent DNA damage response
- PMID: 12679521
- PMCID: PMC153632
- DOI: 10.1073/pnas.0730887100
Evidence that the retroviral DNA integration process triggers an ATR-dependent DNA damage response
Abstract
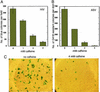
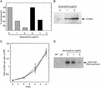
Caffeine is an efficient inhibitor of cellular DNA repair, likely through its effects on ATM (ataxia telangiectasia mutated) and ATR (ATM and Rad3-related) kinases. Here, we show that caffeine treatment causes a dose-dependent reduction in the total amount of HIV-1 and avian sarcoma virus retroviral vector DNA that is joined to host DNA in the population of infected cells and also in the number of transduced cells. These changes were observed at caffeine concentrations that had little or no effect on overall cell growth, synthesis, and nuclear import of the viral DNA, or the activities of the viral integrase in vitro. Substantial reductions in the amount of host-viral-joined DNA in the infected population, and in the number of transductants, were also observed in the presence of a dominant-negative form of the ATR protein, ATRkd. After infection, a significant fraction of these cells undergoes cell death. In contrast, retroviral transduction is not impeded in ATM-deficient cells, and addition of caffeine leads to the same reduction that was observed in ATM-proficient cells. These results suggest that activity of the ATR kinase, but not the ATM kinase, is required for successful completion of the viral DNA integration process and/or survival of transduced cells. Components of the cellular DNA damage repair response may represent potential targets for antiretroviral drug development.
Figures


References
-
- Skalka A M. In: Advances in Virus Research. Maramorosch K, Murphy F A, Shatkin A J, editors. Vol. 52. New York: Academic; 1999. pp. 271–459.
-
- Brin E, Yi J, Skalka A M, Leis J. J Biol Chem. 2000;275:39287–39295. - PubMed
-
- Hazuda D J, Felock P, Witmer M, Wolfe A, Stillmock K, Grobler J A, Espeseth A, Gabryelski L, Schleif W, Blau C, Miller M D. Science. 2000;287:646–650. - PubMed
-
- Pommier Y, Neamati N. Adv Virus Res. 1999;52:427–458. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

