Human immunoglobulin M memory B cells controlling Streptococcus pneumoniae infections are generated in the spleen
- PMID: 12682112
- PMCID: PMC2193885
- DOI: 10.1084/jem.20022020
Human immunoglobulin M memory B cells controlling Streptococcus pneumoniae infections are generated in the spleen
Abstract
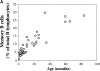
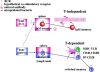
Splenectomized and asplenic patients have a high incidence of infections by encapsulated bacteria and do not respond to polysaccharide vaccines. To understand whether the absence of the spleen is associated with a defined B cell defect, we analyzed B cell subsets in the peripheral blood. We found that a population of B cells known as immunoglobulin (Ig)M memory is lacking in patients without spleen. The absence of IgM memory B cells correlates with an impaired immune response to encapsulated bacteria not only in splenectomized patients, but also in individuals with an intact spleen. We show that the physiological and transient predisposition to pneumococcal infections of young children (0-2 yr) is associated with the lack of circulating IgM memory B cells and of serum antipolysaccharide IgM. We also demonstrate that IgM memory B cells are undetectable in a fraction of patients with common variable immunodeficiency, who have recurrent and invasive infections by encapsulated bacteria. IgM memory B cells, therefore, require the spleen for their generation and/or survival and are responsible for the protection against encapsulated bacteria.
Figures






References
-
- Davidson, R.N., and R.A. Wall. 2001. Prevention and management of infections in patients without a spleen. Clin. Microbiol. Infect. 7:657–660. - PubMed
-
- Feder, H.M., Jr., and H.A. Pearson. 1999. Assessment of splenic function in familial asplenia. N. Engl. J. Med. 341:210–212. - PubMed
-
- Hosea, S.W., C.G. Burch, E.J. Brown, R.A. Berg, and M.M. Frank. 1981. Impaired immune response of splenectomised patients to polyvalent pneumococcal vaccine. Lancet. 1:804–807. - PubMed
-
- Stansfield, S.K. 1987. Acute respiratory infections in the developing world: strategies for prevention, treatment and control. Pediatr. Infect. Dis. J. 6:622–629. - PubMed
-
- Overturf, G.D. 2002. Pneumococcal vaccination of children. Semin. Pediatr. Infect. Dis. 13:155–164. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

