Derangements of hippocampal calcium/calmodulin-dependent protein kinase II in a mouse model for Angelman mental retardation syndrome
- PMID: 12684449
- PMCID: PMC6742065
- DOI: 10.1523/JNEUROSCI.23-07-02634.2003
Derangements of hippocampal calcium/calmodulin-dependent protein kinase II in a mouse model for Angelman mental retardation syndrome
Abstract
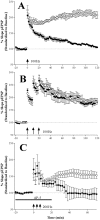
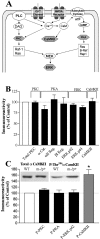
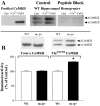
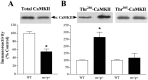
Angelman syndrome (AS) is a disorder of human cognition characterized by severe mental retardation and epilepsy. Recently, a mouse model for AS (Ube3a maternal null mutation) was developed that displays deficits in both context-dependent learning and hippocampal long-term potentiation (LTP). In the present studies, we examined the molecular basis for these LTP and learning deficits. Mutant animals exhibited a significant increase in hippocampal phospho-calcium/calmodulin-dependent protein kinase II (CaMKII), specifically at sites Thr(286) and Thr(305), with no corresponding change in the levels of total CaMKII. In addition, mutants show a reduction in CaMKII activity, autophosphorylation capability, and total CaMKII associated with postsynaptic density. These findings are the first to implicate misregulation of CaMKII as a molecular cause for the neurobehavioral deficits in a human learning disorder.
Figures


Similar articles
-
Translocation of autophosphorylated calcium/calmodulin-dependent protein kinase II to the postsynaptic density.J Biol Chem. 1997 May 23;272(21):13467-70. doi: 10.1074/jbc.272.21.13467. J Biol Chem. 1997. PMID: 9153188
-
CaMKII-dependent phosphorylation of NR2A and NR2B is decreased in animals characterized by hippocampal damage and impaired LTP.Eur J Neurosci. 1999 Jan;11(1):141-8. doi: 10.1046/j.1460-9568.1999.00414.x. Eur J Neurosci. 1999. PMID: 9987018
-
Age-related deficits in long-term potentiation are insensitive to hydrogen peroxide: coincidence with enhanced autophosphorylation of Ca2+/calmodulin-dependent protein kinase II.J Neurosci Res. 2002 Nov 1;70(3):298-308. doi: 10.1002/jnr.10427. J Neurosci Res. 2002. PMID: 12391589
-
Postsynaptic protein phosphorylation and LTP.Trends Neurosci. 2000 Feb;23(2):75-80. doi: 10.1016/s0166-2236(99)01490-3. Trends Neurosci. 2000. PMID: 10652548 Review.
-
CaM kinase II in long-term potentiation.Neurochem Int. 1996 Apr;28(4):343-58. doi: 10.1016/0197-0186(95)00097-6. Neurochem Int. 1996. PMID: 8740440 Review.
Cited by
-
Prader-Willi, Angelman, and 15q11-q13 Duplication Syndromes.Pediatr Clin North Am. 2015 Jun;62(3):587-606. doi: 10.1016/j.pcl.2015.03.004. Epub 2015 Apr 22. Pediatr Clin North Am. 2015. PMID: 26022164 Free PMC article. Review.
-
Synaptic plasticity in mouse models of autism spectrum disorders.Korean J Physiol Pharmacol. 2012 Dec;16(6):369-78. doi: 10.4196/kjpp.2012.16.6.369. Epub 2012 Dec 10. Korean J Physiol Pharmacol. 2012. PMID: 23269898 Free PMC article.
-
UBE3A and Its Link With Autism.Front Mol Neurosci. 2018 Dec 4;11:448. doi: 10.3389/fnmol.2018.00448. eCollection 2018. Front Mol Neurosci. 2018. PMID: 30568575 Free PMC article. Review.
-
Pathophysiological power of improper tonic GABA(A) conductances in mature and immature models.Front Neural Circuits. 2013 Oct 24;7:170. doi: 10.3389/fncir.2013.00170. eCollection 2013. Front Neural Circuits. 2013. PMID: 24167475 Free PMC article. Review.
-
Regulation of CaMKII in vivo: the importance of targeting and the intracellular microenvironment.Neurochem Res. 2009 Oct;34(10):1792-804. doi: 10.1007/s11064-009-9985-9. Epub 2009 May 5. Neurochem Res. 2009. PMID: 19415486 Review.
References
-
- Abel T, Nguyen PV, Barad M, Deuel TA, Kandel ER, Bourtchouladze R. Genetic demonstration of a role for PKA in the late phase of LTP and in hippocampus-based long-term memory. Cell. 1997;88:615–626. - PubMed
-
- Abeliovich A, Chen C, Goda Y, Silva AJ, Stevens CF, Tonegawa S. Modified hippocampal long-term potentiation in PKC-γ-mutant mice. Cell. 1993a;75:1253–1262. - PubMed
-
- Abeliovich A, Paylor R, Chen C, Kim JJ, Wehner JM, Tonegawa S. PKC-γ mutant mice exhibit mild deficits in spatial and contextual learning. Cell. 1993b;75:1263–1271. - PubMed
-
- Adams JP, Sweatt JD. Molecular psychology: roles for the ERK MAP kinase cascade in memory. Annu Rev Pharmacol Toxicol. 2002;42:135–163. - PubMed
-
- Basu A, Haldar S. Signal-induced site specific phosphorylation targets Bcl2 to the proteasome pathway. Int J Oncol. 2002;21:597–601. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
