Different circuits for ON and OFF retinal ganglion cells cause different contrast sensitivities
- PMID: 12684450
- PMCID: PMC6742092
- DOI: 10.1523/JNEUROSCI.23-07-02645.2003
Different circuits for ON and OFF retinal ganglion cells cause different contrast sensitivities
Abstract
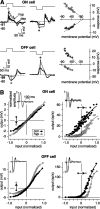
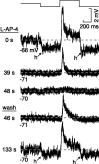
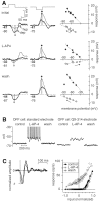
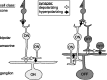
The theory of "parallel pathways" predicts that, except for a sign reversal, ON and OFF ganglion cells are driven by a similar presynaptic circuit. To test this hypothesis, we measured synaptic inputs to ON and OFF cells as reflected in the subthreshold membrane potential. We made intracellular recordings from brisk-transient (Y) cells in the in vitro guinea pig retina and show that ON and OFF cells in fact express significant asymmetries in their synaptic inputs. An ON cell receives relatively linear input that modulates a single excitatory conductance; whereas an OFF cell receives rectified input that modulates both inhibitory and excitatory conductances. The ON pathway, blocked by L-AP-4, tonically inhibits an OFF cell at mean luminance and phasically inhibits an OFF cell during a light increment. Our results suggest that basal glutamate release is high at ON but not OFF bipolar terminals, and inhibition between pathways is unidirectional: ON --> OFF. These circuit asymmetries explain asymmetric contrast sensitivity observed in spiking behavior.
Figures
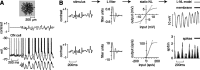


Similar articles
-
Distinct expressions of contrast gain control in parallel synaptic pathways converging on a retinal ganglion cell.J Physiol. 2008 Nov 15;586(22):5487-502. doi: 10.1113/jphysiol.2008.156224. Epub 2008 Oct 2. J Physiol. 2008. PMID: 18832424 Free PMC article.
-
Synaptic pathways that shape the excitatory drive in an OFF retinal ganglion cell.J Neurophysiol. 2012 Apr;107(7):1795-807. doi: 10.1152/jn.00924.2011. Epub 2011 Dec 28. J Neurophysiol. 2012. PMID: 22205648 Free PMC article.
-
Interactions of inhibition and excitation in the light-evoked currents of X type retinal ganglion cells.J Neurophysiol. 1998 Dec;80(6):2975-90. doi: 10.1152/jn.1998.80.6.2975. J Neurophysiol. 1998. PMID: 9862900
-
Diverse inhibitory and excitatory mechanisms shape temporal tuning in transient OFF α ganglion cells in the rabbit retina.J Physiol. 2018 Feb 1;596(3):477-495. doi: 10.1113/JP275195. Epub 2018 Jan 15. J Physiol. 2018. PMID: 29222817 Free PMC article.
-
Functional circuitry for peripheral suppression in Mammalian Y-type retinal ganglion cells.J Neurophysiol. 2007 Jun;97(6):4327-40. doi: 10.1152/jn.01091.2006. Epub 2007 Apr 25. J Neurophysiol. 2007. PMID: 17460102
Cited by
-
Two Pairs of ON and OFF Retinal Ganglion Cells Are Defined by Intersectional Patterns of Transcription Factor Expression.Cell Rep. 2016 May 31;15(9):1930-44. doi: 10.1016/j.celrep.2016.04.069. Epub 2016 May 19. Cell Rep. 2016. PMID: 27210758 Free PMC article.
-
Asymmetric ON-OFF processing of visual motion cancels variability induced by the structure of natural scenes.Elife. 2019 Oct 15;8:e47579. doi: 10.7554/eLife.47579. Elife. 2019. PMID: 31613221 Free PMC article.
-
Spatial segregation of adaptation and predictive sensitization in retinal ganglion cells.Neuron. 2013 Aug 7;79(3):541-54. doi: 10.1016/j.neuron.2013.06.011. Neuron. 2013. PMID: 23932000 Free PMC article.
-
Perspectives on: information and coding in mammalian sensory physiology: nonlinear spatial encoding by retinal ganglion cells: when 1 + 1 ≠ 2.J Gen Physiol. 2011 Sep;138(3):283-90. doi: 10.1085/jgp.201110629. J Gen Physiol. 2011. PMID: 21875977 Free PMC article. No abstract available.
-
Awake responses suggest inefficient dense coding in the mouse retina.Elife. 2023 Nov 3;12:e78005. doi: 10.7554/eLife.78005. Elife. 2023. PMID: 37922200 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
