ADP-ribosylation factor 6 and a functional PIX/p95-APP1 complex are required for Rac1B-mediated neurite outgrowth
- PMID: 12686588
- PMCID: PMC153101
- DOI: 10.1091/mbc.e02-07-0406
ADP-ribosylation factor 6 and a functional PIX/p95-APP1 complex are required for Rac1B-mediated neurite outgrowth
Abstract
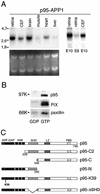
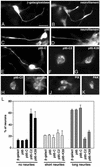
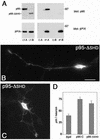
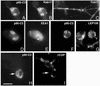
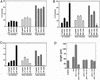
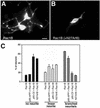
The mechanisms coordinating adhesion, actin organization, and membrane traffic during growth cone migration are poorly understood. Neuritogenesis and branching from retinal neurons are regulated by the Rac1B/Rac3 GTPase. We have identified a functional connection between ADP-ribosylation factor (Arf) 6 and p95-APP1 during the regulation of Rac1B-mediated neuritogenesis. P95-APP1 is an ADP-ribosylation factor GTPase-activating protein (ArfGAP) of the GIT family expressed in the developing nervous system. We show that Arf6 has a predominant role in neurite extension compared with Arf1 and Arf5. Cotransfection experiments indicate a specific and cooperative potentiation of neurite extension by Arf6 and the carboxy-terminal portion of p95-APP1. Localization studies in neurons expressing different p95-derived constructs show a codistribution of p95-APP1 with Arf6, but not Arf1. Moreover, p95-APP1-derived proteins with a mutated or deleted ArfGAP domain prevent Rac1B-induced neuritogenesis, leading to PIX-mediated accumulation at large Rab11-positive endocytic vesicles. Our data support a role of p95-APP1 as a specific regulator of Arf6 in the control of membrane trafficking during neuritogenesis.
Figures


References
-
- Adler R, Jerdan J, Hewitt AT. Responses of cultured neural retinal cells to substratum-bound laminin and other extracellular matrix molecules. Dev Biol. 1985;112:100–114. - PubMed
-
- Bernard V, Pohl BP, Bokoch GM. Characterization of Rac and Cdc42 activation in chemoattractant-stimulated human neutrophils using a novel assay for active GTPases. J Biol Chem. 1999;274:13198–13204. - PubMed
-
- Chomczynski P, Sacchi N. Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal Biochem. 1987;162:156–159. - PubMed
-
- Craig AM, Wyborski RJ, Banker G. Preferential addition of newly synthesized membrane protein at axonal growth cones. Nature. 1995;375:592–594. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

