N-acylethanolamine signaling in tobacco is mediated by a membrane-associated, high-affinity binding protein
- PMID: 12692337
- PMCID: PMC166934
- DOI: 10.1104/pp.102.014936
N-acylethanolamine signaling in tobacco is mediated by a membrane-associated, high-affinity binding protein
Erratum in
- Plant Physiol. 2003 May 21;132(1):400
Abstract
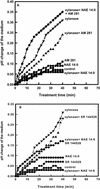
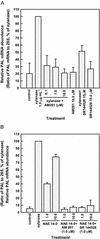
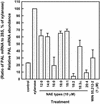
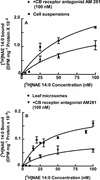
N-Acylethanolamines (NAEs) are fatty acid derivatives found as minor constituents of animal and plant tissues, and their levels increase 10- to 50-fold in tobacco (Nicotiana tabacum) leaves treated with fungal elicitors. Infiltration of tobacco leaves with submicromolar to micromolar concentrations of N-myristoylethanolamine (NAE 14:0) resulted in an increase in relative phenylalanine ammonia-lyase (PAL) transcript abundance within 8 h after infiltration, and this PAL activation was reduced after co-infiltration with cannabinoid receptor antagonists (AM 281 and SR 144528). A saturable, high-affinity specific binding activity for [(3)H]NAE 14:0 was identified in suspension-cultured tobacco cells and in microsomes from tobacco leaves (apparent K(d) of 74 and 35 nM, respectively); cannabinoid receptor antagonists reduced or eliminated specific [(3)H]NAE 14:0 binding, consistent with the physiological response. N-Oleoylethanolamine activated PAL2 expression in leaves and diminished [(3)H]NAE 14:0 binding in microsomes, whereas N-linoleoylethanolamine did not activate PAL2 expression in leaves, and did not affect [(3)H]NAE 14:0 binding in microsomes. The nonionic detergent dodecylmaltoside solubilized functional [(3)H]NAE 14:0-binding activity from tobacco microsomal membranes. The dodecylmaltoside-solubilized NAE-binding activity retained similar, but not identical, binding properties to the NAE-binding protein(s) in intact tobacco microsomes. Additionally, high-affinity saturable NAE-binding proteins were identified in microsomes isolated from Arabidopsis and Medicago truncatula tissues, indicating the general prevalence of these binding proteins in plant membranes. We propose that plants possess an NAE-signaling pathway with functional similarities to the "endocannabinoid" pathway of animal systems and that this pathway, in part, participates in xylanase elicitor perception in tobacco.
Figures
Similar articles
-
N-Acylethanolamines in signal transduction of elicitor perception. Attenuation Of alkalinization response and activation of defense gene expression.Plant Physiol. 1999 Dec;121(4):1299-308. doi: 10.1104/pp.121.4.1299. Plant Physiol. 1999. PMID: 10594117 Free PMC article.
-
Manipulation of Arabidopsis fatty acid amide hydrolase expression modifies plant growth and sensitivity to N-acylethanolamines.Proc Natl Acad Sci U S A. 2006 Aug 8;103(32):12197-202. doi: 10.1073/pnas.0603571103. Epub 2006 Jul 31. Proc Natl Acad Sci U S A. 2006. PMID: 16880402 Free PMC article.
-
Hydrogen peroxide from the oxidative burst is neither necessary nor sufficient for hypersensitive cell death induction, phenylalanine ammonia lyase stimulation, salicylic acid accumulation, or scopoletin consumption in cultured tobacco cells treated with elicitin.Plant Physiol. 1999 Sep;121(1):163-72. doi: 10.1104/pp.121.1.163. Plant Physiol. 1999. PMID: 10482671 Free PMC article.
-
The N-acylethanolamine-mediated regulatory pathway in plants.Chem Biodivers. 2007 Aug;4(8):1933-55. doi: 10.1002/cbdv.200790161. Chem Biodivers. 2007. PMID: 17712835 Review.
-
N-acylethanolamine metabolism with special reference to N-acylethanolamine-hydrolyzing acid amidase (NAAA).Prog Lipid Res. 2010 Oct;49(4):299-315. doi: 10.1016/j.plipres.2010.02.003. Epub 2010 Feb 10. Prog Lipid Res. 2010. PMID: 20152858 Review.
Cited by
-
Differential effects of two phospholipase D inhibitors, 1-butanol and N-acylethanolamine, on in vivo cytoskeletal organization and Arabidopsis seedling growth.Protoplasma. 2005 Dec;226(3-4):109-23. doi: 10.1007/s00709-005-0124-4. Epub 2005 Dec 12. Protoplasma. 2005. PMID: 16333570
-
N-Acylethanolamine metabolism interacts with abscisic acid signaling in Arabidopsis thaliana seedlings.Plant Cell. 2007 Aug;19(8):2454-69. doi: 10.1105/tpc.106.048702. Epub 2007 Aug 31. Plant Cell. 2007. PMID: 17766402 Free PMC article.
-
Phospholipase D and phosphatidic acid in plant defence response: from protein-protein and lipid-protein interactions to hormone signalling.J Exp Bot. 2015 Apr;66(7):1721-36. doi: 10.1093/jxb/eru540. Epub 2015 Feb 13. J Exp Bot. 2015. PMID: 25680793 Free PMC article. Review.
-
The hrpN gene of Erwinia amylovora stimulates tobacco growth and enhances resistance to Botrytis cinerea.Planta. 2006 Feb;223(3):449-56. doi: 10.1007/s00425-005-0100-4. Epub 2005 Sep 1. Planta. 2006. PMID: 16136336
References
-
- Anderson JD, Bailey BA, Dean JFD, Taylor R. A fungal endoxylanase elicits ethylene biosynthesis in tobacco (Nicotiana tabacum L. cv. Xanthi) leaves. In: Flores HE, Arteca RN, Shannon JC, editors. Polyamines and Ethylene: Biochemistry, Physiology, and Interactions. Rockville, MD: American Society of Plant Physiologists; 1990. pp. 146–156.
-
- Baker CJ, O'Neill NR, Keppler LD, Orlandi EW. Early responses during plant-bacterial interactions in tobacco cell suspensions. Phytopathology. 1991;81:1504–1507.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

