Amyloid deposition in the hippocampus and entorhinal cortex: quantitative analysis of a transgenic mouse model
- PMID: 12697936
- PMCID: PMC153642
- DOI: 10.1073/pnas.0330745100
Amyloid deposition in the hippocampus and entorhinal cortex: quantitative analysis of a transgenic mouse model
Abstract
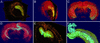
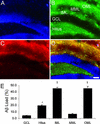
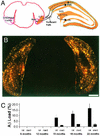
Various transgenic mouse models of Alzheimer's disease (AD) have been developed that overexpress mutant forms of amyloid precursor protein in an effort to elucidate more fully the potential role of beta-amyloid (A beta) in the etiopathogenesis of the disease. The present study represents the first complete 3D reconstruction of A beta in the hippocampus and entorhinal cortex of PDAPP transgenic mice. A beta deposits were detected by immunostaining and thioflavin fluorescence, and quantified by using high-throughput digital image acquisition and analysis. Quantitative analysis of amyloid load in hippocampal subfields showed a dramatic increase between 12 and 15 months of age, with little or no earlier detectable deposition. Three-dimensional reconstruction in the oldest brains visualized previously unrecognized sheets of A beta coursing through the hippocampus and cerebral cortex. In contrast with previous hypotheses, compact plaques form before significant deposition of diffuse A beta, suggesting that different mechanisms are involved in the deposition of diffuse amyloid and the aggregation into plaques. The dentate gyrus was the hippocampal subfield with the greatest amyloid burden. Sublaminar distribution of A beta in the dentate gyrus correlated most closely with the termination of afferent projections from the lateral entorhinal cortex, mirroring the selective vulnerability of this circuit in human AD. This detailed temporal and spatial analysis of A beta and compact amyloid deposition suggests that specific corticocortical circuits express selective, but late, vulnerability to the pathognomonic markers of amyloid deposition, and can provide a basis for detecting prior vulnerability factors.
Figures


References
-
- Price D L, Sisodia S S. Annu Rev Neurosci. 1998;21:479–505. - PubMed
-
- Hardy J, Selkoe D J. Science. 2002;297:353–356. - PubMed
-
- Sommer B. Curr Opin Pharmacol. 2002;2:87–92. - PubMed
-
- Masliah E, Rockenstein E. J Neural Transm (Suppl.) 2000;59:175–183. - PubMed
-
- Dodart J C, Mathis C, Saura J, Bales K R, Paul S M, Ungerer A. Neurobiol Dis. 2000;7:71–85. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

