Use of the Galleria mellonella caterpillar as a model host to study the role of the type III secretion system in Pseudomonas aeruginosa pathogenesis
- PMID: 12704110
- PMCID: PMC153283
- DOI: 10.1128/IAI.71.5.2404-2413.2003
Use of the Galleria mellonella caterpillar as a model host to study the role of the type III secretion system in Pseudomonas aeruginosa pathogenesis
Abstract
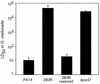
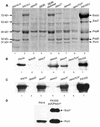
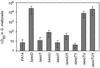
Nonvertebrate model hosts represent valuable tools for the study of host-pathogen interactions because they facilitate the identification of bacterial virulence factors and allow the discovery of novel components involved in host innate immune responses. In this report, we determined that the greater wax moth caterpillar Galleria mellonella is a convenient nonmammalian model host for study of the role of the type III secretion system (TTSS) in Pseudomonas aeruginosa pathogenesis. Based on the observation that a mutation in the TTSS pscD gene of P. aeruginosa strain PA14 resulted in a highly attenuated virulence phenotype in G. mellonella, we examined the roles of the four known effector proteins of P. aeruginosa (ExoS, ExoT, ExoU, and ExoY) in wax moth killing. We determined that in P. aeruginosa strain PA14, only ExoT and ExoU play a significant role in G. mellonella killing. Strain PA14 lacks the coding sequence for the ExoS effector protein and does not seem to express ExoY. Moreover, using Delta exoU Delta exoY, Delta exoT Delta exoY, and Delta exoT Delta exoU double mutants, we determined that individual translocation of either ExoT or ExoU is sufficient to obtain nearly wild-type levels of G. mellonella killing. On the other hand, data obtained with a Delta exoT Delta exoU Delta exoY triple mutant and a Delta pscD mutant suggested that additional, as-yet-unidentified P. aeruginosa components of type III secretion are involved in virulence in G. mellonella. A high level of correlation between the results obtained in the G. mellonella model and the results of cytopathology assays performed with a mammalian tissue culture system validated the use of G. mellonella for the study of the P. aeruginosa TTSS.
Figures
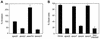

Similar articles
-
The exoS, exoT, exoU and exoY Virulotypes of the Type 3 Secretion System in Multidrug Resistant Pseudomonas aeruginosa as a Death Risk Factor in Pediatric Patients.Pathogens. 2024 Nov 22;13(12):1030. doi: 10.3390/pathogens13121030. Pathogens. 2024. PMID: 39770290 Free PMC article.
-
ExoU is a potent intracellular phospholipase.Mol Microbiol. 2004 Sep;53(5):1279-90. doi: 10.1111/j.1365-2958.2004.04194.x. Mol Microbiol. 2004. PMID: 15387809 Review.
-
Prevalence of type III secretion genes in clinical and environmental isolates of Pseudomonas aeruginosa.Microbiology (Reading). 2001 Oct;147(Pt 10):2659-2669. doi: 10.1099/00221287-147-10-2659. Microbiology (Reading). 2001. PMID: 11577145
-
Alveolar response to Pseudomonas aeruginosa: role of the type III secretion system.Infect Immun. 2005 Jul;73(7):4263-71. doi: 10.1128/IAI.73.7.4263-4271.2005. Infect Immun. 2005. PMID: 15972518 Free PMC article.
-
Role of Pseudomonas aeruginosa type III effectors in disease.Curr Opin Microbiol. 2009 Feb;12(1):61-6. doi: 10.1016/j.mib.2008.12.007. Epub 2009 Jan 23. Curr Opin Microbiol. 2009. PMID: 19168385 Review.
Cited by
-
Mitigation of the Toxic Effects of Periodontal Pathogens by Candidate Probiotics in Oral Keratinocytes, and in an Invertebrate Model.Front Microbiol. 2020 Jun 16;11:999. doi: 10.3389/fmicb.2020.00999. eCollection 2020. Front Microbiol. 2020. PMID: 32612578 Free PMC article.
-
Role of acinetobactin-mediated iron acquisition functions in the interaction of Acinetobacter baumannii strain ATCC 19606T with human lung epithelial cells, Galleria mellonella caterpillars, and mice.Infect Immun. 2012 Mar;80(3):1015-24. doi: 10.1128/IAI.06279-11. Epub 2012 Jan 9. Infect Immun. 2012. PMID: 22232188 Free PMC article.
-
Traversal of multilayered corneal epithelia by cytotoxic Pseudomonas aeruginosa requires the phospholipase domain of exoU.Invest Ophthalmol Vis Sci. 2012 Jan 25;53(1):448-53. doi: 10.1167/iovs.11-8999. Invest Ophthalmol Vis Sci. 2012. PMID: 22167094 Free PMC article.
-
Novel type III effectors in Pseudomonas aeruginosa.mBio. 2015 Mar 17;6(2):e00161. doi: 10.1128/mBio.00161-15. mBio. 2015. PMID: 25784698 Free PMC article.
-
Impact of AmpC Derepression on Fitness and Virulence: the Mechanism or the Pathway?mBio. 2016 Oct 25;7(5):e01783-16. doi: 10.1128/mBio.01783-16. mBio. 2016. PMID: 27795406 Free PMC article.
References
-
- Aballay, A., and F. M. Ausubel. 2002. Caenorhabditis elegans as a host for the study of host-pathogen interactions. Curr. Opin. Microbiol. 5:97-101. - PubMed
-
- Ausubel, F. M., R. Brent, R. E. Kingston, D. D. Moore, J. G. Seidman, J. A. Smith, and K. Struhl (ed.). 1992. Short protocols in molecular biology, 2nd ed. John Wiley & Sons, Inc., New York, N.Y.
-
- Cohn, J., G. Sessa, and G. B. Martin. 2001. Innate immunity in plants. Curr. Opin. Immunol. 13:55-62. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

