Peroxisome proliferator-activated receptor alpha is involved in the regulation of lipid metabolism by ginseng
- PMID: 12711630
- PMCID: PMC1573779
- DOI: 10.1038/sj.bjp.0705169
Peroxisome proliferator-activated receptor alpha is involved in the regulation of lipid metabolism by ginseng
Abstract
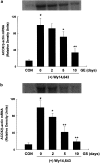
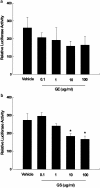
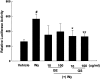
1. Peroxisome proliferator-activated receptor alpha (PPARalpha) regulates the expression of the key genes involved in lipid metabolism following activation of this receptor by various ligands. Ginseng, a highly valuable medicine in oriental societies, is also reported to modulate lipid metabolism, although the mechanism of its action remains unknown. In order to test our hypothesis that ginseng exerts its effects by altering PPARalpha-mediated pathways, the effects of Korean red ginseng on PPARalpha function and serum lipid profiles were investigated using in vivo and in vitro approaches. 2. In vivo administration of ginseng extract (GE) and ginsenosides (GS) not only inhibited mRNA levels of acyl-CoA oxidase, a rate-limiting enzyme for PPARalpha-mediated peroxisomal fatty acid beta-oxidation, induced by the potent PPARalpha ligand Wy14,643 in a dose- and time-dependent manner, but also inhibited the induction of PPARalpha target genes expected following treatment with Wy14,643. 3. Consistent with the in vivo data, both GE and GS caused dose-dependent decreases in the endogenous expression of a luciferase reporter gene containing the PPAR responsive element (PPRE), while GS significantly decreased the magnitude of reporter gene activation in the presence of Wy14,643. 4. Serological studies demonstrated that, compared with vehicle-treated mice, treatment with GS significantly increased serum concentrations of total cholesterol, triglycerides, and high-density lipoprotein (HDL) cholesterol. Compared to groups treated with Wy14,643 alone, which significantly decreased serum triglyceride and HDL cholesterol levels versus controls, coadministration of either GE or GS with Wy14,643 modestly increased serum triglycerides and HDL cholesterol. 5. These results indicate that the effects of ginseng on serum lipid profiles may be mediated by changes in the expression of PPARalpha target genes, providing the first evidence that in vivo and in vitro treatments of ginseng modulate PPARalpha action. In addition, these data suggest that ginseng can act as an inhibitor of PPARalpha function, which may have therapeutic implications.
Figures



Similar articles
-
Ginsenoside Rf, a component of ginseng, regulates lipoprotein metabolism through peroxisome proliferator-activated receptor alpha.Biochem Biophys Res Commun. 2006 Jan 6;339(1):196-203. doi: 10.1016/j.bbrc.2005.10.197. Epub 2005 Nov 10. Biochem Biophys Res Commun. 2006. PMID: 16297877
-
Inhibition of the actions of peroxisome proliferator-activated receptor alpha on obesity by estrogen.Obesity (Silver Spring). 2007 Jun;15(6):1430-40. doi: 10.1038/oby.2007.171. Obesity (Silver Spring). 2007. PMID: 17557980
-
Importance of PPAR alpha for the effects of growth hormone on hepatic lipid and lipoprotein metabolism.Growth Horm IGF Res. 2007 Apr;17(2):154-64. doi: 10.1016/j.ghir.2007.01.003. Epub 2007 Feb 20. Growth Horm IGF Res. 2007. PMID: 17307376
-
Molecular mechanisms of alcoholic fatty liver: role of peroxisome proliferator-activated receptor alpha.Alcohol. 2004 Aug;34(1):35-8. doi: 10.1016/j.alcohol.2004.07.005. Alcohol. 2004. PMID: 15670663 Review.
-
Peroxisome proliferator-activated receptors and atherogenesis: regulators of gene expression in vascular cells.Circ Res. 2004 May 14;94(9):1168-78. doi: 10.1161/01.RES.0000127122.22685.0A. Circ Res. 2004. PMID: 15142970 Review.
Cited by
-
Traditional chinese medicine in treatment of metabolic syndrome.Endocr Metab Immune Disord Drug Targets. 2008 Jun;8(2):99-111. doi: 10.2174/187153008784534330. Endocr Metab Immune Disord Drug Targets. 2008. PMID: 18537696 Free PMC article. Review.
-
Effects of ginseng on two main sex steroid hormone receptors: estrogen and androgen receptors.J Ginseng Res. 2017 Apr;41(2):215-221. doi: 10.1016/j.jgr.2016.08.005. Epub 2016 Aug 23. J Ginseng Res. 2017. PMID: 28413327 Free PMC article. Review.
-
The study of ginsenoside on PPARgamma expression of mononuclear macrophage in type 2 diabetes.Mol Biol Rep. 2010 Jul;37(6):2975-9. doi: 10.1007/s11033-009-9864-0. Epub 2009 Oct 10. Mol Biol Rep. 2010. PMID: 19821054
-
Effects of Korean white ginseng extracts on obesity in high-fat diet-induced obese mice.Cytotechnology. 2010 Aug;62(4):367-76. doi: 10.1007/s10616-010-9288-7. Epub 2010 Sep 23. Cytotechnology. 2010. PMID: 20862608 Free PMC article.
-
The Efficacy of Ginseng (Panax) on Human Prediabetes and Type 2 Diabetes Mellitus: A Systematic Review and Meta-Analysis.Nutrients. 2022 Jun 9;14(12):2401. doi: 10.3390/nu14122401. Nutrients. 2022. PMID: 35745129 Free PMC article.
References
-
- AOYAMA T., PETERS J.M., IRITANI N., NAKAJIMA T., FURIHATA K., HASHIMOTO T., GONZALEZ F.J. Altered constitutive expression of fatty acid-metabolizing enzymes in mice lacking the peroxisome proliferator-activated receptor α (PPARα) J. Biol. Chem. 1998;273:5678–5684. - PubMed
-
- ATTELE A.S., WU J.A., YUAN C.S. Ginseng pharmacology: multiple constituents and multiple actions. Biochem. Pharmacol. 1999;58:1685–1693. - PubMed
-
- AUWERX J., SCHOONJANS K., FRUCHART J.C., STAELS B. Transcriptional control of triglyceride metabolism: fibrates and fatty acids change the expression of the LPL and apo C-III genes by activating the nuclear receptor PPAR. Atherosclerosis. 1996;124 Suppl:S29–S37. - PubMed
-
- BANTHORPE D.V.Terpenoids Natural Products 1994Essex: Longman Scientific and Technical; 331–339.ed. Mann, J. pp
-
- CHUNG E., LEE K.Y., LEE Y.J., LEE Y.H., LEE S.K. Ginsenoside Rg1 down-regulates glucocorticoid receptor and displays synergistic effects with cAMP. Steroids. 1998;63:421–424. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

