Noradrenergic neurons in the rat solitary nucleus participate in the esophageal-gastric relaxation reflex
- PMID: 12714355
- PMCID: PMC3062485
- DOI: 10.1152/ajpregu.00155.2003
Noradrenergic neurons in the rat solitary nucleus participate in the esophageal-gastric relaxation reflex
Abstract
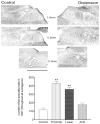
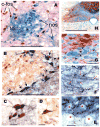
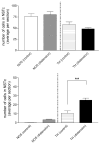
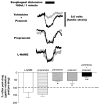
Activation of esophageal mechanosensors excites neurons in and near the central nucleus of the solitary tract (NSTc). In turn, NSTc neurons coordinate the relaxation of the stomach [i.e., the receptive relaxation reflex (RRR)] by modulating the output of vagal efferent neurons of the dorsal motor nucleus of the vagus (DMN). The NSTc area contains neurons with diverse neurochemical phenotypes, including a large population of catecholaminergic and nitrergic neurons. The aim of the present study was to determine whether either one of these prominent neuronal phenotypes was involved in the RRR. Immunohistochemical techniques revealed that repetitive esophageal distension caused 53% of tyrosine hydroxylase-immunoreactive (TH-ir) neurons to colocalize c-Fos in the NSTc. No nitric oxide synthase (NOS)-ir neurons in the NSTc colocalized c-Fos in either distension or control conditions. Local brain stem application (2 ng) of alpha-adrenoreceptor antagonists (i.e., alpha1-prazosin or alpha2-yohimbine) significantly reduced the magnitude of the esophageal distension-induced gastric relaxation to approximately 55% of control conditions. The combination of yohimbine and prazosin reduced the magnitude of the reflex to approximately 27% of control. In contrast, pretreatment with either the NOS-inhibitor NG-nitro-l-arginine methyl ester or the beta-adrenoceptor antagonist propranolol did not interfere with esophageal distension-induced gastric relaxation. Unilateral microinjections of the agonist norepinephrine (0.3 ng) directed at the DMN were sufficient to mimic the transient esophageal-gastric reflex. Our data suggest that noradrenergic, but not nitrergic, neurons of the NSTc play a prominent role in the modulation of the RRR through action on alpha1- and alpha2-adrenoreceptors. The finding that esophageal afferent stimulation alone is not sufficient to activate NOS-positive neurons in the NSTc suggests that these neurons may be strongly gated by other central nervous system inputs, perhaps related to the coordination of swallowing or emesis with respiration.
Figures


Similar articles
-
Hindbrain chemical mediators of reflex-induced inhibition of gastric tone produced by esophageal distension and intravenous nicotine.Am J Physiol Regul Integr Comp Physiol. 2005 Nov;289(5):R1482-95. doi: 10.1152/ajpregu.00003.2005. Epub 2005 Jul 28. Am J Physiol Regul Integr Comp Physiol. 2005. PMID: 16051723
-
Esophageal-gastric relaxation reflex in rat: dual control of peripheral nitrergic and cholinergic transmission.Am J Physiol Regul Integr Comp Physiol. 2006 Jun;290(6):R1570-6. doi: 10.1152/ajpregu.00717.2005. Epub 2006 Jan 26. Am J Physiol Regul Integr Comp Physiol. 2006. PMID: 16439669 Free PMC article.
-
Brainstem pathways responsible for oesophageal control of gastric motility and tone in the rat.J Physiol. 1999 Jan 15;514 ( Pt 2)(Pt 2):369-83. doi: 10.1111/j.1469-7793.1999.369ae.x. J Physiol. 1999. PMID: 9852320 Free PMC article.
-
Neural circuits in swallowing and abdominal vagal afferent-mediated lower esophageal sphincter relaxation.Am J Med. 2001 Dec 3;111 Suppl 8A:95S-105S. doi: 10.1016/s0002-9343(01)00863-4. Am J Med. 2001. PMID: 11749933 Review.
-
Central control of lower esophageal sphincter relaxation.Am J Med. 2000 Mar 6;108 Suppl 4a:90S-98S. doi: 10.1016/s0002-9343(99)00345-9. Am J Med. 2000. PMID: 10718459 Review.
Cited by
-
Inhibitory neurotransmission regulates vagal efferent activity and gastric motility.Exp Biol Med (Maywood). 2016 Jun;241(12):1343-50. doi: 10.1177/1535370216654228. Exp Biol Med (Maywood). 2016. PMID: 27302177 Free PMC article. Review.
-
Hindbrain noradrenergic lesions attenuate anorexia and alter central cFos expression in rats after gastric viscerosensory stimulation.J Neurosci. 2003 Nov 5;23(31):10084-92. doi: 10.1523/JNEUROSCI.23-31-10084.2003. J Neurosci. 2003. PMID: 14602823 Free PMC article.
-
Short-term receptor trafficking in the dorsal vagal complex: an overview.Auton Neurosci. 2006 Jun 30;126-127:2-8. doi: 10.1016/j.autneu.2006.01.019. Epub 2006 Mar 6. Auton Neurosci. 2006. PMID: 16580267 Free PMC article. Review.
-
Advances in the physiology of gastric emptying.Neurogastroenterol Motil. 2019 Apr;31(4):e13546. doi: 10.1111/nmo.13546. Epub 2019 Feb 10. Neurogastroenterol Motil. 2019. PMID: 30740834 Free PMC article. Review.
-
Hindbrain noradrenergic A2 neurons: diverse roles in autonomic, endocrine, cognitive, and behavioral functions.Am J Physiol Regul Integr Comp Physiol. 2011 Feb;300(2):R222-35. doi: 10.1152/ajpregu.00556.2010. Epub 2010 Oct 20. Am J Physiol Regul Integr Comp Physiol. 2011. PMID: 20962208 Free PMC article. Review.
References
-
- Altschuler SM, Bao XM, Bieger D, Hopkins DA, Miselis RR. Viscerotopic representation of the upper alimentary tract in the rat: sensory ganglia and nuclei of the solitary and spinal trigeminal tracts. J Comp Neurol. 1989;283:248–268. - PubMed
-
- Bertolino M, Vicini S, Gillis R, Travagli A. Presynaptic α2-adrenoceptors inhibit excitatory synaptic transmission in rat brain stem. Am J Physiol Gastrointest Liver Physiol. 1997;272:G654–G661. - PubMed
-
- Bieger D. The brainstem esophagomotor network pattern generator: a rodent model. Dysphagia. 1993;8:203–208. - PubMed
-
- Bieger D, Hopkins DA. Viscerotopic representation of the upper alimentary tract in the medulla oblongata in the rat: the nucleus ambiguus. J Comp Neurol. 1987;262:546–562. - PubMed
-
- Blessing WW. The Lower Brainstem and Bodily Homeostasis. Oxford, UK: Oxford Univ. Press; 1997. Eating and metabolism; pp. 323–372.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

