Ligand-induced internalization of the p75 neurotrophin receptor: a slow route to the signaling endosome
- PMID: 12716928
- PMCID: PMC6742322
- DOI: 10.1523/JNEUROSCI.23-08-03209.2003
Ligand-induced internalization of the p75 neurotrophin receptor: a slow route to the signaling endosome
Abstract
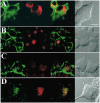
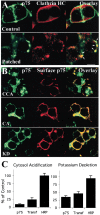
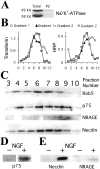
The nerve growth factor (NGF) family of neurotrophins binds two classes of cell-surface receptors, trk receptor tyrosine kinases and the shared p75 receptor. Rapid internalization and retrograde trafficking of neurotrophin-trk complexes have been demonstrated in a number of systems and are thought to transmit trophic signals from terminals to neuronal cell bodies. In contrast, the internalization and trafficking of neurotrophin-p75 complexes are not well understood. In this study, we used biotinylated NGF and a fluorescent-labeled anti-p75 antibody to follow the kinetics and route of ligand-induced internalization of the p75 receptor in cycling and differentiated PC12 cells. Binding of neurotrophins to p75 induced internalization at a rate approximately three times slower than that of transferrin and NGF-TrkA complexes in the same cells. The ligand-p75 complex was internalized via clathrin-coated pits into early endosomes and eventually accumulated in recycling endosomes in the cell body and vesicles colabeled by the cholera toxin B-subunit in the growth cones. Both internalized ligand and p75 were protected from proteolytic degradation and accumulated in vesicles that did not undergo acidification. Finally, NGF induced endosomal association of p75 and its MAGE interactors, necdin and NRAGE. These data suggest that signaling endosomes containing activated p75 are involved in neurotrophin signaling, and that such endosomes may be temporally and spatially distinct from those containing trk receptors.
Figures






References
-
- Barker PA. p75NTR: a study in contrasts. Cell Death Differ. 1998;5:346–356. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
