Autocrine/paracrine activation of the GABA(A) receptor inhibits the proliferation of neurogenic polysialylated neural cell adhesion molecule-positive (PSA-NCAM+) precursor cells from postnatal striatum
- PMID: 12716935
- PMCID: PMC6742317
- DOI: 10.1523/JNEUROSCI.23-08-03278.2003
Autocrine/paracrine activation of the GABA(A) receptor inhibits the proliferation of neurogenic polysialylated neural cell adhesion molecule-positive (PSA-NCAM+) precursor cells from postnatal striatum
Abstract
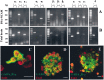
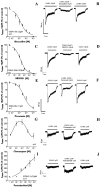
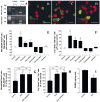
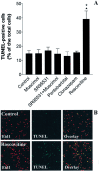
GABA and its type A receptor (GABA(A)R) are present in the immature CNS and may function as growth-regulatory signals during the development of embryonic neural precursor cells. In the present study, on the basis of their isopycnic properties in a buoyant density gradient, we developed an isolation procedure that allowed us to purify proliferative neural precursor cells from early postnatal rat striatum, which expressed the polysialylated form of the neural cell adhesion molecule (PSA-NCAM). These postnatal striatal PSA-NCAM+ cells were shown to proliferate in the presence of epidermal growth factor (EGF) and formed spheres that preferentially generated neurons in vitro. We demonstrated that PSA-NCAM+ neuronal precursors from postnatal striatum expressed GABA(A)R subunits in vitro and in situ. GABA elicited chloride currents in PSA-NCAM+ cells by activation of functional GABA(A)R that displayed a typical pharmacological profile. GABA(A)R activation in PSA-NCAM+ cells triggered a complex intracellular signaling combining a tonic inhibition of the mitogen-activated protein kinase cascade and an increase of intracellular calcium concentration by opening of voltage-gated calcium channels. We observed that the activation of GABA(A)R in PSA-NCAM+ neuronal precursors from postnatal striatum inhibited cell cycle progression both in neurospheres and in organotypic slices. Furthermore, postnatal PSA-NCAM+ striatal cells synthesized and released GABA, thus creating an autocrine/paracrine mechanism that controls their proliferation. We showed that EGF modulated this autocrine/paracrine loop by decreasing GABA production in PSA-NCAM+ cells. This demonstration of GABA synthesis and GABA(A)R function in striatal PSA-NCAM+ cells may shed new light on the understanding of key extrinsic cues that regulate the developmental potential of postnatal neuronal precursors in the CNS.
Figures





Similar articles
-
Control of cell survival and proliferation of postnatal PSA-NCAM(+) progenitors.Mol Cell Neurosci. 2003 Feb;22(2):162-78. doi: 10.1016/s1044-7431(02)00030-1. Mol Cell Neurosci. 2003. PMID: 12676527
-
Regulation of the polysialylated form of the neural cell adhesion molecule in the developing striatum: effects of cortical lesions.J Comp Neurol. 1997 Dec 15;389(2):289-308. doi: 10.1002/(sici)1096-9861(19971215)389:2<289::aid-cne8>3.0.co;2-y. J Comp Neurol. 1997. PMID: 9416923
-
Growth and fate of PSA-NCAM+ precursors of the postnatal brain.J Neurosci. 1998 Aug 1;18(15):5777-88. doi: 10.1523/JNEUROSCI.18-15-05777.1998. J Neurosci. 1998. PMID: 9671666 Free PMC article.
-
Untangling the functional potential of PSA-NCAM-expressing cells in CNS development and brain repair strategies.Curr Med Chem. 2003 Oct;10(20):2185-96. doi: 10.2174/0929867033456774. Curr Med Chem. 2003. PMID: 12871092 Review.
-
PSA-NCAM in mammalian structural plasticity and neurogenesis.Prog Neurobiol. 2006 Oct;80(3):129-64. doi: 10.1016/j.pneurobio.2006.08.003. Epub 2006 Oct 9. Prog Neurobiol. 2006. PMID: 17029752 Review.
Cited by
-
Diabetes Impairs Wnt3 Protein-induced Neurogenesis in Olfactory Bulbs via Glutamate Transporter 1 Inhibition.J Biol Chem. 2016 Jul 15;291(29):15196-211. doi: 10.1074/jbc.M115.672857. Epub 2016 May 20. J Biol Chem. 2016. PMID: 27226528 Free PMC article.
-
Effect of midazolam on the proliferation of neural stem cells isolated from rat hippocampus.Neural Regen Res. 2012 Jul 5;7(19):1475-82. doi: 10.3969/j.issn.1673-5374.2012.19.005. Neural Regen Res. 2012. PMID: 25657682 Free PMC article.
-
Emerging intersections between neuroscience and glioma biology.Nat Neurosci. 2019 Dec;22(12):1951-1960. doi: 10.1038/s41593-019-0540-y. Epub 2019 Nov 12. Nat Neurosci. 2019. PMID: 31719671 Review.
-
GABA(A) Increases Calcium in Subventricular Zone Astrocyte-Like Cells Through L- and T-Type Voltage-Gated Calcium Channels.Front Cell Neurosci. 2010 Apr 8;4:8. doi: 10.3389/fncel.2010.00008. eCollection 2010. Front Cell Neurosci. 2010. PMID: 20422045 Free PMC article.
-
Kinetic properties of the alpha2 homo-oligomeric glycine receptor impairs a proper synaptic functioning.J Physiol. 2003 Dec 1;553(Pt 2):369-86. doi: 10.1113/jphysiol.2003.052142. Epub 2003 Sep 12. J Physiol. 2003. PMID: 12972628 Free PMC article.
References
-
- Barker JL, Behar T, Li YX, Liu QY, Ma W, Maric D, Maric I, Schaffner AE, Serafini R, Smith SV, Somogyi R, Vautrin JY, Wen XL, Xian H. GABAergic cells and signals in CNS development. Perspect Dev Neurobiol. 1998;5:305–322. - PubMed
-
- Belachew S, Malgrange B, Rigo JM, Rogister B, Leprince P, Hans G, Nguyen L, Moonen G. Glycine triggers an intracellular calcium influx in oligodendrocyte progenitor cells which is mediated by the activation of both the ionotropic glycine receptor and Na+-dependent transporters. Eur J Neurosci. 2000;12:1924–1930. - PubMed
-
- Bettendorff L, Sallanon-Moulin M, Touret M, Wins P, Margineanu I, Schoffeniels E. Paradoxical sleep deprivation increases the content of glutamate and glutamine in rat cerebral cortex. Sleep. 1996;19:65–71. - PubMed
-
- Bettendorff L, Lakaye B, Margineanu I, Grisar T, Wins P. ATP-driven, NA+-independent inward Cl- pumping in neuroblastoma cells. J Neurochem. 2002;81:792–801. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous
