Functional NMDA receptor subtype 2B is expressed in astrocytes after ischemia in vivo and anoxia in vitro
- PMID: 12716944
- PMCID: PMC6742326
- DOI: 10.1523/JNEUROSCI.23-08-03364.2003
Functional NMDA receptor subtype 2B is expressed in astrocytes after ischemia in vivo and anoxia in vitro
Abstract
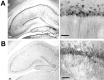
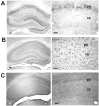
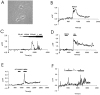
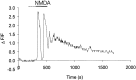
NMDA-type glutamate receptors play a critical role in neuronal synaptogenesis, plasticity, and excitotoxic death. Recent studies indicate that functional NMDA receptors are also expressed in certain glial populations in the normal brain. Using immunohistochemical methods, we detected the presence of the NMDA receptor 2B (NR2B) subunit of the NMDA receptor in neurons but not astrocytes in the CA1 and subicular regions of the rat hippocampus. However, after ischemia-induced neuronal death in these regions, double immunohistochemical labeling revealed that NR2B subunits colocalized with the astrocyte marker glial fibrillary acid protein and with NR1 subunits that are required for functional NMDA receptors. NR2B expression was first observed 3 d after ischemia and reached a peak at 28 d. At 56 d, only a few NR2B-expressing astrocytes were still present. In vitro, when postnatal hippocampal cultures were subjected to 5 min of anoxia, it resulted in NR2B expression on astrocytes in the glial feed layer. Imaging of intracellular calcium with postanoxic cultures and astrocytes isolated acutely from the ischemic hippocampus revealed a rise in intracellular [Ca2+] after stimulation with the specific agonist NMDA. The response could be blocked reversibly with the competitive antagonist 2-amino-5-phosphonovalerate and attenuated by the NR2B-selective antagonist ifenprodil. Control astrocytes were not responsive to NMDA but responded to glutamate. An understanding of the role of astrocytes that express functional NMDA receptors in response to ischemia may guide development of novel stroke therapies.
Figures




References
-
- Abdel-Hamid KM, Baimbridge KG. The effects of artificial calcium buffers on calcium responses and glutamate-mediated excitotoxicity in cultured hippocampal neurons. Neuroscience. 1997;81:673–687. - PubMed
-
- Amédée T, Robert A, Coles JA. Potassium homeostasis and glial energy metabolism. Glia. 1997;21:46–55. - PubMed
-
- Anderson CM, Bridges RJ, Chamberlin AR, Shimamoto K, Yasuda-Kamatami Y, Swanson RA. Differing effects of substrate and non-substrate transport inhibitors on glutamate uptake reversal. J Neurochem. 2001;79:1207–1216. - PubMed
-
- Angelov DN, Neiss WF. Neuronal recovery after peripheral traumatic lesions of the facial motor nerve. Biomed Rev. 1994;3:39–53.
-
- Arabadzisz D, Freund TF. Changes in excitatory and inhibitory circuits of the rat hippocampus 12–14 months after complete forebrain ischemia. Neuroscience. 1999;92:27–45. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous
