Cooperative regulation of myosin-actin interactions by a continuous flexible chain II: actin-tropomyosin-troponin and regulation by calcium
- PMID: 12719246
- PMCID: PMC1302877
- DOI: 10.1016/S0006-3495(03)70041-1
Cooperative regulation of myosin-actin interactions by a continuous flexible chain II: actin-tropomyosin-troponin and regulation by calcium
Abstract
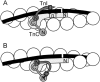
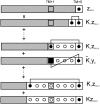
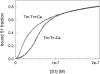
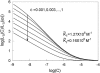
The model of myosin regulation by a continuous tropomyosin chain is generalized to a chain of tropomyosin-troponin units. Myosin binding to regulated actin is cooperative and initially inhibited by the chain as before. In the absence of calcium, myosin is further inhibited by the binding of troponin-I to actin, which through the whole of troponin pins the tropomyosin chain in a blocking position; myosin and TnI compete for actin and induce oppositely-directed chain kinks. The model predicts equilibrium binding curves for myosin-S1 and TnI as a function of their first-order affinities K(S1) and L(TI). Myosin is detached by the actin binding of TnI, but TnI is more efficiently detached by myosin when the kink size (typically nine to ten actin sites) spans the seven-site spacing between adjacent TnI molecules. An allosteric mechanism is used for coupling the detachment of TnI to calcium binding by TnC. With thermally activated TnI kinks (kink energy B approximately k(B)T), TnI also binds cooperatively to actin, producing cooperative detachment of myosin and biphasic myosin-calcium Hill plots, with Hill coefficients of 2 at high calcium and 4-6 at low calcium as observed in striated muscle. The theory also predicts the cooperative effects observed in the calcium loading of TnC.
Figures




Similar articles
-
Cooperative regulation of myosin-actin interactions by a continuous flexible chain I: actin-tropomyosin systems.Biophys J. 2003 May;84(5):3155-67. doi: 10.1016/S0006-3495(03)70040-X. Biophys J. 2003. PMID: 12719245 Free PMC article.
-
Cooperative regulation of myosin-S1 binding to actin filaments by a continuous flexible Tm-Tn chain.Eur Biophys J. 2012 Dec;41(12):1015-32. doi: 10.1007/s00249-012-0859-8. Epub 2012 Oct 7. Eur Biophys J. 2012. PMID: 23052974 Free PMC article.
-
Structural basis for Ca2+-regulated muscle relaxation at interaction sites of troponin with actin and tropomyosin.J Mol Biol. 2005 Sep 9;352(1):178-201. doi: 10.1016/j.jmb.2005.06.067. J Mol Biol. 2005. PMID: 16061251
-
Structural basis for calcium-regulated relaxation of striated muscles at interaction sites of troponin with actin and tropomyosin.Adv Exp Med Biol. 2007;592:71-86. doi: 10.1007/978-4-431-38453-3_8. Adv Exp Med Biol. 2007. PMID: 17278357 Review.
-
Regulation of contraction in striated muscle.Physiol Rev. 2000 Apr;80(2):853-924. doi: 10.1152/physrev.2000.80.2.853. Physiol Rev. 2000. PMID: 10747208 Review.
Cited by
-
Age-Onset Phosphorylation of a Minor Actin Variant Promotes Intestinal Barrier Dysfunction.Dev Cell. 2019 Dec 2;51(5):587-601.e7. doi: 10.1016/j.devcel.2019.11.001. Dev Cell. 2019. PMID: 31794717 Free PMC article.
-
Cooperativity of myosin II motors in the non-regulated and regulated thin filaments investigated with high-speed AFM.J Gen Physiol. 2023 Mar 6;155(3):e202213190. doi: 10.1085/jgp.202213190. Epub 2023 Jan 12. J Gen Physiol. 2023. PMID: 36633585 Free PMC article.
-
Striated muscle regulation of isometric tension by multiple equilibria.PLoS One. 2009 Dec 8;4(12):e8052. doi: 10.1371/journal.pone.0008052. PLoS One. 2009. PMID: 19997610 Free PMC article.
-
The effect of variable troponin C mutation thin filament incorporation on cardiac muscle twitch contractions.J Mol Cell Cardiol. 2021 Jun;155:112-124. doi: 10.1016/j.yjmcc.2021.02.009. Epub 2021 Feb 24. J Mol Cell Cardiol. 2021. PMID: 33636222 Free PMC article.
-
The myosin duty ratio tunes the calcium sensitivity and cooperative activation of the thin filament.Biochemistry. 2013 Sep 17;52(37):6437-44. doi: 10.1021/bi400262h. Epub 2013 Sep 3. Biochemistry. 2013. PMID: 23947752 Free PMC article.
References
-
- Bremel, R. D., and A. Weber. 1972. Cooperation within actin filament in vertebrate skeletal muscle. Nat. New Biol. 238:97–101. - PubMed
-
- Ebashi, S. 1977. Troponin and its function. In Search and Discovery: A Tribute to Albert Szent-Gyorgi. B. Kaminer, editor. Academic Press, New York.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

