Development of a LightCycler PCR assay for detection and quantification of Aspergillus fumigatus DNA in clinical samples from neutropenic patients
- PMID: 12734210
- PMCID: PMC154665
- DOI: 10.1128/JCM.41.5.1811-1818.2003
Development of a LightCycler PCR assay for detection and quantification of Aspergillus fumigatus DNA in clinical samples from neutropenic patients
Abstract
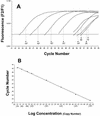
The increasing incidence of invasive aspergillosis, a life-threatening infection in immunocompromised patients, emphasizes the need to improve the diagnostic tools for this disease. We established a LightCycler-based real-time PCR assay to detect and quantify rapidly, specifically, and sensitively Aspergillus fumigatus DNA in both bronchoalveolar lavage (BAL) and blood samples from high-risk patients. The primers and hybridization probes were derived from an A. fumigatus-specific sequence of the mitochondrial cytochrome b gene. The assay is linear in the range between 13.2 fg and 1.3 ng of A. fumigatus DNA, corresponding to 3 to 300,000 CFU per ml of BAL fluid or blood. No cross-amplification was observed with human DNA or with the DNA of fungal or bacterial pathogens. For clinical evaluation we investigated 10 BAL samples from nine neutropenic patients with malignant hematological diseases and 12 blood samples from seven neutropenic patients with malignant hematological diseases. Additionally, we tested one blood sample and one BAL sample from each of two neutropenic patients. In order to characterize the validity of the novel PCR assay, only samples that had shown positive results by a previously described sensitive and specific nested PCR assay were tested. Twelve of 12 BAL samples and 6 of 14 blood samples gave positive results by the LightCycler PCR assay. Eight of 14 blood samples gave negative results by the novel method. The LightCycler PCR-mediated quantification of the fungal burden showed 15 to 269,018 CFU per ml of BAL sample and 298 to 104,114 CFU per ml of blood sample. Twenty of 20 BAL samples and 50 of 50 blood samples from subjects without evidence of invasive pulmonary aspergillosis (IPA) were PCR negative. Compared to a previously described nested PCR assay, these preliminary data for the novel real-time PCR assay indicate a less sensitive rate of detection of IPA in high-risk patients, but the assay may be valuable for quantification of the fungal burden in individual clinical samples.
Figures

References
-
- Anonymous. 1994. Recommendations for diagnostic bronchoalveolar lavage. German Society of Pneumology. Pneumologie 48(Suppl. 1):311-323. - PubMed
-
- Ascioglu, S., J. H. Rex, B. de Pauw, J. E. Bennett, J. Bille, F. Crokaert, D. W. Denning, J. P. Donnelly, J. E. Edwards, Z. Erjavec, D. Fiere, O. Lortholary, J. Maertens, J. F. Meis, T. F. Patterson, J. Ritter, D. Selleslag, P. M. Shah, D. A. Stevens, and T. J. Walsh. 2002. Defining opportunistic invasive fungal infections in immunocompromised patients with cancer and hematopoietic stem cell transplants: an international consensus. Clin. Infect. Dis. 34:7-14. - PubMed
-
- Bart-Delabesse, E., J. Sarfati, J. P. Debeaupuis, W. van Leeuwen, A. van Belkum, S. Bretagne, and J. P. Latge. 2001. Comparison of restriction fragment length polymorphism, microsatellite length polymorphism, and random amplification of polymorphic DNA analyses for fingerprinting Aspergillus fumigatus isolates. J. Clin. Microbiol. 39:2683-2686. - PMC - PubMed
-
- Beck-Sague, C., and W. R. Jarvis. 1993. Secular trends in the epidemiology of nosocomial fungal infections in the United States, 1980-1990. National Nosocomial Infections Surveillance System. J. Infect. Dis. 167:1247-1251. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

