Biophysical and pharmacological characterization of voltage-gated calcium currents in turtle auditory hair cells
- PMID: 12740421
- PMCID: PMC2342991
- DOI: 10.1113/jphysiol.2002.037481
Biophysical and pharmacological characterization of voltage-gated calcium currents in turtle auditory hair cells
Abstract
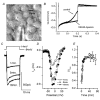
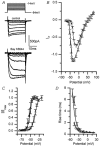
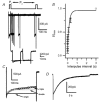
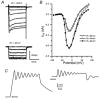
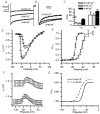
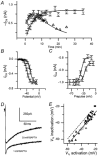
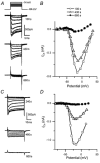
Hair cell calcium channels regulate membrane excitability and control synaptic transmission. The present investigations focused on determining whether calcium channels vary between hair cells of different characteristic frequencies or if multiple channel types exist within a hair cell, each serving a different function. To this end, turtle auditory hair cells from high- (317 +/- 27 Hz) and low-frequency (115 +/- 6 Hz) positions were voltage clamped using the whole-cell recording technique, and calcium currents were characterized based on activation, inactivation and pharmacological properties. Pharmacological sensitivity to dihydropyridines (nimodipine, Bay K 8644), benzothiazepines (diltiazem) and acetonitrile derivatives (verapamil, D600) and the insensitivity to non-L-type calcium channel antagonists support the conclusion that only L-type calcium channels were present. Fast activation rise times (< 0.5 ms), hyperpolarized half-activation potentials and a relative insensitivity to nimodipine suggest the channels were of the alpha1D (CaV1.3) variety. Although no pharmacological differences were found between calcium currents obtained from high- and low-frequency cells, low-frequency cells activated slightly faster and at hyperpolarized potentials, with half-activating voltages of -43 +/- 1 mV compared to -35 +/- 1 mV. Inactivation was observed in both high- and low-frequency cells. The time course of inactivation required three time constants for a fit. Long depolarizations could result in complete inactivation. The voltage of half-inactivation was -40 +/- 2 mV for high-frequency cells and -46 +/- 2 mV for low-frequency cells. Calcium channel inactivation did not significantly alter hair cell electrical resonant properties elicited from protocols where the membrane potential was hyperpolarized or depolarized prior to characterizing the resonance. A bell-shaped voltage dependence and modest sensitivities to intracellular calcium chelators and external barium ions suggest that inactivation was calcium dependent.
Figures


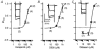

References
-
- Art JJ, Crawford AC, Fettiplace R. Electrical resonance and membrane currents in turtle cochlear hair cells. Hear Res. 1986;22:31–36. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

