Transcription of endogenous and exogenous R2 elements in the rRNA gene locus of Drosophila melanogaster
- PMID: 12748285
- PMCID: PMC155226
- DOI: 10.1128/MCB.23.11.3825-3836.2003
Transcription of endogenous and exogenous R2 elements in the rRNA gene locus of Drosophila melanogaster
Abstract
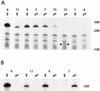
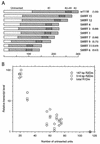
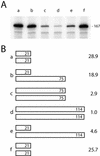
R2 retrotransposons insert into the rRNA-encoding units (rDNA units) that form the nucleoli of insects. We have utilized an R2 integration system in Drosophila melanogaster to study transcription of foreign sequences integrated into the R2 target site of the 28S rRNA genes. The exogenous sequences were cotranscribed at dramatically different levels which closely paralleled the level of transcription of the endogenous R1 and R2 elements. Transcription levels were inversely correlated with the number of uninserted rDNA units, variation in this number having been brought about by the R2 integration system itself. Females with as few as 20 uninserted rDNA units per X chromosome had expression levels of endogenous and exogenous insertion sequences that were 2 orders of magnitude higher than lines that contained over 80 uninserted rDNA units per chromosome. R2 insertions only 167 bp in length exhibited this range of transcriptional regulation. Analysis of transcript levels in males suggested R2 insertions on the Y chromosome are not down-regulated to the same extent as insertions on the X chromosome. These results suggest that transcription of the rDNA units can be tightly regulated, but this regulation gradually breaks down as the cell approaches the minimum number of uninserted genes needed for survival.
Figures




Similar articles
-
Integration, Regulation, and Long-Term Stability of R2 Retrotransposons.Microbiol Spectr. 2015 Apr;3(2):MDNA3-0011-2014. doi: 10.1128/microbiolspec.MDNA3-0011-2014. Microbiol Spectr. 2015. PMID: 26104703 Free PMC article. Review.
-
Chromatin structure and transcription of the R1- and R2-inserted rRNA genes of Drosophila melanogaster.Mol Cell Biol. 2006 Dec;26(23):8781-90. doi: 10.1128/MCB.01409-06. Epub 2006 Sep 25. Mol Cell Biol. 2006. PMID: 17000772 Free PMC article.
-
R1 and R2 retrotransposition and deletion in the rDNA loci on the X and Y chromosomes of Drosophila melanogaster.Genetics. 2003 Oct;165(2):675-85. doi: 10.1093/genetics/165.2.675. Genetics. 2003. PMID: 14573479 Free PMC article.
-
Monitoring the mode and tempo of concerted evolution in the Drosophila melanogaster rDNA locus.Genetics. 2005 Dec;171(4):1837-46. doi: 10.1534/genetics.105.047670. Epub 2005 Sep 2. Genetics. 2005. PMID: 16143606 Free PMC article.
-
Evolution of R1 and R2 in the rDNA units of the genus Drosophila.Genetica. 1997;100(1-3):49-61. Genetica. 1997. PMID: 9440258 Review.
Cited by
-
Integration, Regulation, and Long-Term Stability of R2 Retrotransposons.Microbiol Spectr. 2015 Apr;3(2):MDNA3-0011-2014. doi: 10.1128/microbiolspec.MDNA3-0011-2014. Microbiol Spectr. 2015. PMID: 26104703 Free PMC article. Review.
-
Origin of nascent lineages and the mechanisms used to prime second-strand DNA synthesis in the R1 and R2 retrotransposons of Drosophila.Genome Biol. 2009;10(5):R49. doi: 10.1186/gb-2009-10-5-r49. Epub 2009 May 5. Genome Biol. 2009. PMID: 19416522 Free PMC article.
-
Chromatin structure and transcription of the R1- and R2-inserted rRNA genes of Drosophila melanogaster.Mol Cell Biol. 2006 Dec;26(23):8781-90. doi: 10.1128/MCB.01409-06. Epub 2006 Sep 25. Mol Cell Biol. 2006. PMID: 17000772 Free PMC article.
-
R1 and R2 retrotransposition and deletion in the rDNA loci on the X and Y chromosomes of Drosophila melanogaster.Genetics. 2003 Oct;165(2):675-85. doi: 10.1093/genetics/165.2.675. Genetics. 2003. PMID: 14573479 Free PMC article.
-
Evolutionary dynamics of 5S rDNA location in acridid grasshoppers and its relationship with H3 histone gene and 45S rDNA location.Genetica. 2011 Jul;139(7):921-31. doi: 10.1007/s10709-011-9596-7. Epub 2011 Jul 14. Genetica. 2011. PMID: 21755328
References
-
- Burke, W. D., H. S. Malik, W. C. Lathe, and T. H. Eickbush. 1998. Are retrotransposons long term hitchhikers? Nature 239:141-142. - PubMed
-
- Burke, W. D., H. S. Malik, J. P. Jones, and T. H. Eickbush. 1999. The domain structure and retrotransposition mechanism of R2 elements are conserved throughout arthropods. Mol. Biol. Evol. 16:502-511. - PubMed
-
- Chooi, W. Y. 1979. The occurrence of long transcription units among the X and Y ribosomal genes of Drosophila melanogaster: transcription of insertion sequences. Chromosoma 74:57-74. - PubMed
-
- Clark, A. G., F. M. Szumski, and E. S. M. Lyckegaard. 1990. Population genetics of the Y chromosome of Drosophila melanogaster: rDNA variation and phenotypic correlates. Genet. Res. 58:7-13. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
