Heterozygous deficiency of hypoxia-inducible factor-2alpha protects mice against pulmonary hypertension and right ventricular dysfunction during prolonged hypoxia
- PMID: 12750401
- PMCID: PMC155039
- DOI: 10.1172/JCI15496
Heterozygous deficiency of hypoxia-inducible factor-2alpha protects mice against pulmonary hypertension and right ventricular dysfunction during prolonged hypoxia
Abstract
Chronic hypoxia induces pulmonary vascular remodeling, leading to pulmonary hypertension, right ventricular hypertrophy, and heart failure. Heterozygous deficiency of hypoxia-inducible factor-1alpha (HIF-1alpha), which mediates the cellular response to hypoxia by increasing expression of genes involved in erythropoiesis and angiogenesis, has been previously shown to delay hypoxia-induced pulmonary hypertension. HIF-2alpha is a homologue of HIF-1alpha and is abundantly expressed in the lung, but its role in pulmonary hypertension remains unknown. Therefore, we analyzed the pulmonary response of WT and viable heterozygous HIF-2alpha-deficient (Hif2alpha(+/-)) mice after exposure to 10% O(2) for 4 weeks. In contrast to WT mice, Hif2alpha(+/-) mice were fully protected against pulmonary hypertension and right ventricular hypertrophy, unveiling a critical role of HIF-2alpha in hypoxia-induced pulmonary vascular remodeling. Pulmonary expression levels of endothelin-1 and plasma catecholamine levels were increased threefold and 12-fold respectively in WT but not in Hif2alpha(+/-) mice after hypoxia, suggesting that HIF-2alpha-mediated upregulation of these vasoconstrictors contributes to the development of hypoxic pulmonary vascular remodeling.
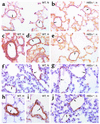
Figures

References
-
- Barbera JA, Peinado VI, Santos S. Pulmonary hypertension in COPD: old and new concepts. Monaldi Arch. Chest Dis. 2000;55:445–449. - PubMed
-
- Hida W, Tun Y, Kikuchi Y, Okabe S, Shirato K. Pulmonary hypertension in patients with chronic obstructive pulmonary disease: recent advances in pathophysiology and management. Respirology. 2002;7:3–13. - PubMed
-
- Barnes PJ. Chronic obstructive pulmonary disease. N. Engl. J. Med. 2000;343:269–280. - PubMed
-
- Chen YF, Oparil S. Endothelin and pulmonary hypertension. J. Cardiovasc. Pharmacol. 2000;35(4 Suppl. 2):S49–S53. - PubMed
-
- Durmowicz AG, Stenmark KR. Mechanisms of structural remodeling in chronic pulmonary hypertension. Pediatr. Rev. 1999;20:e91–e102. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

