Activation of presynaptic glycine receptors facilitates glycine release from presynaptic terminals synapsing onto rat spinal sacral dorsal commissural nucleus neurons
- PMID: 12754315
- PMCID: PMC2343048
- DOI: 10.1113/jphysiol.2003.041053
Activation of presynaptic glycine receptors facilitates glycine release from presynaptic terminals synapsing onto rat spinal sacral dorsal commissural nucleus neurons
Abstract
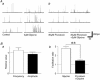
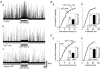
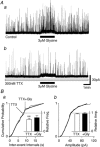
Glycine is a major inhibitory neurotransmitter in the spinal cord and brainstem. Here we report the novel finding that presynaptic glycine autoreceptors modulate release from terminals synapsing onto rat spinal sacral dorsal commissural nucleus (SDCN) neurons. In mechanically dissociated SDCN neurons, in which functional presynaptic nerve terminals remain adherent to the isolated neurons, exogenously applied glycine (3 microM) increased the frequency of glycinergic spontaneous inhibitory postsynaptic currents (sIPSCs) without affecting their amplitudes or decay times. This suggests that glycine acts presynaptically to increase glycine release probability. Picrotoxin, at a concentration that had little direct effect on sIPSC frequency and amplitude (30 microM), significantly attenuated glycine-induced presynaptic sIPSC facilitation. The glycine-induced sIPSC frequency facilitation was completely abolished either in a Ca(2+)-free external solution or in the presence of 100 microM Cd2+, suggesting the involvement of extracellular Ca2+ influx into the nerve terminals. The glycine action was also completely occluded in the presence of 300 nM tetrodotoxin. In recordings from SDCN neurons in spinal cord slices, glycine (10 microM) increased evoked IPSC (eIPSC) amplitude and decreased the extent of paired-pulse facilitation. In response to brief high frequency stimulus trains the eIPSCs displayed a profound frequency-dependent facilitation that was greatly reduced by picrotoxin (30 microM). These results indicate that glycine acts at presynaptic autoreceptors, causing depolarization of the glycinergic nerve terminals, the subsequent activation of voltage-dependent Na+ and Ca2+ channels, and facilitation of glycine release. Furthermore, this presynaptic facilitation was observed under more physiological conditions, suggesting that these glycinergic autoreceptors may contribute to the integration of local inhibitory inputs to SDCN neurons.
Figures

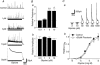

Similar articles
-
Differential modulation of evoked and spontaneous glycine release from rat spinal cord glycinergic terminals by the cyclic AMP/protein kinase A transduction cascade.J Neurochem. 2004 Nov;91(3):657-66. doi: 10.1111/j.1471-4159.2004.02741.x. J Neurochem. 2004. PMID: 15485496
-
Effects of various K+ channel blockers on spontaneous glycine release at rat spinal neurons.Brain Res. 2007 Jul 9;1157:11-22. doi: 10.1016/j.brainres.2006.09.097. Epub 2007 Jun 6. Brain Res. 2007. PMID: 17555723
-
The effect of zinc on glycinergic inhibitory postsynaptic currents in rat spinal dorsal horn neurons.Brain Res. 2007 Aug 3;1161:11-20. doi: 10.1016/j.brainres.2007.05.060. Epub 2007 Jun 12. Brain Res. 2007. PMID: 17604007
-
Regulation of excitation by glycine receptors.Results Probl Cell Differ. 2008;44:123-43. doi: 10.1007/400_2007_029. Results Probl Cell Differ. 2008. PMID: 17541751 Review.
-
The inhibitory neuronal glycine receptor.Bioessays. 1994 Oct;16(10):735-44. doi: 10.1002/bies.950161008. Bioessays. 1994. PMID: 7980477 Review.
Cited by
-
Presynaptic glycine receptors as a potential therapeutic target for hyperekplexia disease.Nat Neurosci. 2014 Feb;17(2):232-9. doi: 10.1038/nn.3615. Epub 2014 Jan 5. Nat Neurosci. 2014. PMID: 24390226 Free PMC article.
-
Differential distribution of glycine receptor subtypes at the rat calyx of Held synapse.J Neurosci. 2012 Nov 21;32(47):17012-24. doi: 10.1523/JNEUROSCI.1547-12.2012. J Neurosci. 2012. PMID: 23175852 Free PMC article.
-
Control of presynaptic function by a persistent Na(+) current.Neuron. 2008 Dec 26;60(6):975-9. doi: 10.1016/j.neuron.2008.10.052. Neuron. 2008. PMID: 19109905 Free PMC article.
-
Presynaptic glycine receptors increase GABAergic neurotransmission in rat periaqueductal gray neurons.Neural Plast. 2013;2013:954302. doi: 10.1155/2013/954302. Epub 2013 Sep 1. Neural Plast. 2013. PMID: 24078885 Free PMC article.
-
Glycine receptors and glycine transporters: targets for novel analgesics?Cell Mol Life Sci. 2018 Feb;75(3):447-465. doi: 10.1007/s00018-017-2622-x. Epub 2017 Aug 8. Cell Mol Life Sci. 2018. PMID: 28791431 Free PMC article. Review.
References
-
- Akaike N, Harata N. Nystatin perforated patch recording and its application to analysis of intracellular mechanism. Jpn J Physiol. 1994;44:433–473. - PubMed
-
- Akaike N, Moorhouse AJ. Techniques: Applications of the nerve-bouton preparation in neuropharmacology. Trends Pharmacol Sci. 2003;24:44–47. - PubMed
-
- Bohlhalter S, Mohler H, Fritschy JM. Inhibitory neurotransmission in rat spinal cord: co-localization of glycine- and GABAA-receptors at GABAergic synaptic contacts demonstrated by triple immunofluorescence staining. Brain Res. 1994;642:59–69. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

